Best Management Practices for Managing Herbicide Resistance
Start Clean
Use agronomic practices that limit the introduction and spread of weeds. In other words: Prevent weed problems before they start.
Seed into weed-free fields and keep them weed-free.
One of the fundamental ways to prevent development of herbicide-resistant weeds is by controlling all weeds prior to seeding a crop and by keeping fields clean (weed-free) throughout the growing season. Because herbicide resistance is thought to be present at very low frequencies (for example, one plant in a million) before selection pressure from herbicides are applied, keeping weed numbers low reduces the chance of that rare resistant plant being present in your field.
Remove or cut weeds before seed set.
Reduce the soil seedbank (the reserve of viable seeds present on the soil surface and scattered throughout the soil profile) by removing or destroying any weed seed that is produced. Preventing weeds from setting seeds may not benefit the current crop but will reduce the weed seedbank over time. The old saying “One year’s seeding, seven years’ weeding” may be even more important when talking about herbicide-resistant weeds because tools to manage those biotypes might be very limited.
Manage weed seed at harvest and after harvest to prevent weed seedbank buildup.
Practices to control weed seed at harvest have proved to be useful to reduce the soil seedbank, particularly of potential herbicide-resistant weeds that survive in-crop herbicide applications. Examples of some of these practices are the bale direct systems, chaff lining and chaff tramlining, or impact mills integrated into combines to destroy the weed seeds in the chaff (see Harvest Weed Seed Control: Applications for PNW Wheat Production Systems [Lyon et al. 2019]). Harvest weed seed control practices target the control of species that retain a significant amount of seeds at crop maturity such as tumble mustard or common lambsquarters. However, for species that set seed after harvest, like Russian thistle or prickly lettuce, it is very important to control these weeds after harvest to prevent larger infestations in those fields and neighboring fields into which they may move or disperse seed.
Compost livestock manure.
Applying composted livestock manure to fields can be a good way to improve soil organic matter and provide beneficial nutrients. However, manure can also be a source of weed infestations. Manure should be correctly composted to ensure sufficient heating to kill weed seeds. Turn and mix the compost pile to ensure all the potential weed seeds are exposed to high temperatures (130–145 degrees Fahrenheit).
Use weed-free seed.
Seeding crop seed contaminated with weed seeds has been the most common method of spreading weeds for centuries. To minimize this risk, use high-quality, weed-free seed, or purchase certified seed.
Understand weed biology, particularly timing of seed germination, seed dormancy, and seed longevity.
Although practices to either break down seed dormancy or encourage seed germination to reduce the soil seedbank are not going to be a “silver bullet,” they will help to both decrease the probability of having herbicide resistance problems and reduce overall weed pressure. An understanding of the conditions that favor or limit germination (to manage seed longevity and seed dormancy) in problematic weed species will help to optimize herbicide application timings and agronomic practices (seeding dates, residue management, tillage practices, crop rotations) that will lead to improved control.
Prevent field-to-field and within-field movement by starting equipment usage in weed-free areas and by cleaning equipment after use.
Prevention is the most effective method of dealing with weeds. Restrict the opportunity for new weeds to invade and spread. Do not allow machinery or vehicles to enter your property unless they are clean. Restrict the movement of vehicles and machinery on your property during periods when seeds are likely to spread. If a weed infestation already exists in a field, start working from the clean areas, and leave the infested areas for last. Clean the equipment after operating it in infested areas, particularly harvesting equipment.
Control weeds in borders to prevent weed influx into the field.
Weeds in unmanaged areas, including field margins, roadsides, rights-of-way, and ditch banks, can serve as a source for the introduction and movement of new weed species including herbicide-resistant weeds. Allowing weed seed production in field borders can have long-term effects on the seedbank, especially when outcrossing occurs with resistant populations near a field, allowing the spread of resistance through pollen and seed movement.
Stay Clean
Help the crop compete with weeds. In other words: Help the crop “choke out” weeds.
Use crop competitiveness and other cultural management strategies, including crop rotation.
Prepare a firm, moist seedbed.
Seed-to-soil contact is key to maximizing germination potential. Ensure row cleaners, packing wheels, and other seeder attachments are correctly adjusted to gently firm the soil without overly displacing it or excessively packing it. Seeding into adequate, not excessive, soil moisture promotes quicker germination and lowers some disease pressures. Manage crop residue to avoid “hair-pinning” straw while seeding.
Seed at an optimum time for rapid germination and emergence.
Delay seeding until soil temperatures and moisture are optimal for crop emergence. Avoid cool and excessively wet seeding conditions to maximize even stand development and minimize opportunities for weeds to emerge (Table 1).
Table 1. Soil temperatures for rapid crop germination.
| Crop | Minimum (°F) | Optimal (°F) |
|---|---|---|
| Wheat | 37 | 54–77a |
| Spring Barley | 40 | 55–75b |
| Dry Field Pea | 40 | 60–70c |
| Chickpea | 41 | 50–59d |
| Sp. Canola | 41 | 50e |
Sources: aEvans et al. (1975); bStark (2003); cOelke et al. (1991); dCorp et al. (2004); eOplinger et al. (1989).
Select competitive crop cultivars.
Choose varieties that emerge quickly, grow rapidly, and swiftly form a crop canopy. In addition, varieties with robust disease tolerances to known site problems will minimize risks of uneven stand development.
Use high-quality seed.
Choose larger seed with a sufficient test weight and known germination potential from a trusted source, such as certified seed. Avoid seed with excessive small, shriveled kernels, even if it has a good test weight. Plump seeds emerge quickly and will be more resilient in adverse seeding conditions.
Use treated seed.
Seed treated with appropriate fungicides and insecticides is protected from pests that can slow early growth and cause stand loss that results in reduced competition with weeds.
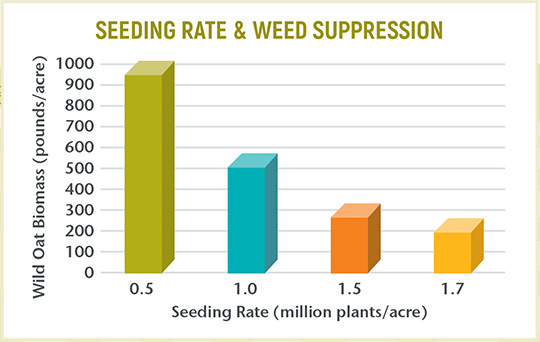
Use higher seeding rates.
Higher seeding rates cause plants to be more competitive for resources providing better weed suppression through competition (Figure 1).
Figure 1. Wild oat biomass in spring barley at different seeding rates in late June.
Use narrowest feasible row spacing.
Narrower row spacing allows crops to form a canopy more quickly and suppress weeds earlier in the season.
Seed on the shallow side of the recommended seeding depth.
Shallower seeding results in faster seedling emergence and leads to earlier establishment of the crop canopy to suppress weed emergence.
Apply fertilizer to promote crop growth and competitiveness.
Do a soil test to determine optimal fertility for your yield goals. Apply fertilizer in bands below the seed when possible to maximize fertility for the crop and minimize resource competition from weeds. Use a starter fertilizer to maximize seedling emergence speed and early season vigor.
Remain Clean
Use practices that keep weeds “off balance.” In other words: Do not allow weeds to adapt.
Diversify weed management practices.
Rotate crops with varied life cycles and seeding dates.
Winter annual weeds that germinate during late fall can be controlled before seeding spring crops. Fall-seeded crops compete well with annual weeds that germinate during late spring or summer. In general, more herbicides are available to control grass weeds in broadleaf crops or to control broadleaf weeds in grass crops. Crops that grow taller rather than shorter, crops that are seeded in the winter rather than spring, and crops that tiller higher and faster (e.g., barley versus wheat) often compete against weeds better. Incorporation of a perennial crop, especially a forage, is competitive against annual weeds. Increase the number and diversity of crops and the length of the rotation.
Rotate or combine herbicides with different mechanisms of action (MOA) and activity on each weed species.
Rotating or combining MOA helps only when all herbicides in the rotation or tank mix have activity on the target weed species. Following the initial herbicide treatments, subsequent herbicides can be used to control resistant plants that have emerged.
Rotate use of herbicide-resistant crops.
The use of herbicide-resistant crops allows for increased herbicide rotation and often control of a weed species that has no other herbicide management options. Avoid overuse of one type of herbicide-resistant crop. For example, do not use Clearfield or CoAXium wheat more than once in three years, and always use them in systems that include preemergence herbicides.
Rotate tillage practices.
Annual grasses and perennial weeds tend to predominate in no-till systems, whereas annual broadleaf weeds tend to flourish in tilled systems. Occasionally changing tillage practices can prevent one or more weed species from proliferating in a field. A light harrow or cultivation of the soil can induce weed seed germination and allow subsequent control with herbicides. Herbicide rotation can be expanded by using herbicides that require mechanical incorporation.
Incorporate cover crops.
Cover crops delay or prevent weed seed germination and compete with weeds that do grow.
Use multiple herbicide mechanisms of action that are effective on troublesome or herbicide-resistant weeds.
Research has shown that applying two herbicides that provide good control of the same weed species will reduce the likelihood of herbicide resistance. They must have a different mechanism of action and a different metabolic breakdown process in the plant. They should be applied in a way that minimizes antagonism. This could include a preemergence herbicide plus a postemergence herbicide if both herbicides will still be active at the same time. Weed seed production will likely be lower when herbicides are combined rather than just rotated.
Follow the herbicide label.
Use the correct rate at recommended weed sizes. Reduced herbicide rates, or application to larger than recommended weeds, might increase the incidence of resistance. Optimize spray application timing, carrier volume, and adjuvant use for better control.
Scout fields routinely.
Closely monitor which weeds are present and the outcome of herbicide treatments. This will tell you which weed species are in the field, where they are, and the severity of the infestation. Proper weed identification is key. Scout early and scout often! Most weeds are easier to control when small. Herbicides rarely work against mature weeds. Scout before seeding to ensure crops are planted into a weed-free field and soon after seeding to assess the efficacy of treatments and decide on further treatments. Take note if the area of a single weed species is spreading, if an herbicide application failed on a single weed, and if some weeds of the same species remained alive while others died. Contact your local university or Extension educator if resistant weeds are suspected or if you need help with weed identification.
Know and understand the effects of weed management inputs on each weed species!
Resources
Table 2 summarizes how the choice of management options influences the chance for developing herbicide resistance in a weed species. Use it to help you assess how likely your management practices are to result in the selection of herbicide-resistant weed biotypes on your farm.
Table 2. Risk of resistance on a per species basis.
| Management Option | Low Risk of Resistance | Moderate Risk of Resistance | High Risk of Resistance |
|---|---|---|---|
| Herbicide mix or rotation in cropping system | More than 2 mechanisms of action | 2 mechanisms of action | 1 mechanism of action |
| Weed control in cropping system | Cultural, mechanical, and chemical | Cultural and chemical | Chemical alone |
| Use of same mechanism of action per season | Once | More than once | Many times |
| Cropping system | Full rotation | Limited rotation | No rotation |
| Resistance status to mechanism of action | Unknown | Limited | Common |
| Weed infestation | Low | Moderate | High |
| Control in last three years | Good | Declining | Poor |
Adapted from A Herbicide Resistance Risk Matrix (Moss et al. 2019).
Use the list to indicate those integrated weed management practices you are already using and those that you intend to implement in the current or coming season. Try to identify at least one new management practice listed under each of the three integrated weed management principles that you will try to implement.
Best management practices (BMPs) for managing herbicide resistance on your farm.
Use agronomic practices that limit the introduction and spread of weeds
- Remove or cut weeds before seed set—Already Doing—Will Implement
- Manage weed seed at harvest and after harvest—Already Doing—Will Implement
- Compost livestock manure—Already Doing—Will Implement
- Use weed-free seed—Already Doing—Will Implement
- Understand weed biology—Already Doing—Will Implement
- Prevent field-to-field and within field movement—Already Doing—Will Implement
- Control weeds in borders—Already Doing—Will Implement
Help the crop compete with weeds
- Prepare a firm, moist seedbed—Already Doing—Will Implement
- Seed at optimum time—Already Doing—Will Implement
- Select competitive crop cultivars—Already Doing—Will Implement
- Use high quality seed—Already Doing—Will Implement
- Use treated seed—Already Doing—Will Implement
- Use higher seeding rates—Already Doing—Will Implement
- Plant on the shallow side of the recommended seeding depth—Already Doing—Will Implement
- Apply fertilizer to promote crop growth and competitiveness—Already Doing—Will Implement
Use practices that keep weeds “off balance”
- Rotate crops with varied life cycles and seeding dates—Already Doing—Will Implement
- Rotate or combine herbicides with different mechanisms of action—Already Doing—Will Implement
- Rotate use of herbicide-resistant crops—Already Doing—Will Implement
- Rotate tillage practices—Already Doing—Will Implement
- Incorporate cover crops—Already Doing—Will Implement
- Use multiple herbicide mechanisms of action—Already Doing—Will Implement
- Follow the herbicide label—Already Doing—Will Implement
- Scout fields routinely—Already Doing—Will Implement
- Know and understand the effects of weed management inputs—Already Doing—Will Implement
MANAGING HERBICIDE-RESISTANT WEEDS IN THE PACIFIC NORTHWEST
Best Management Practices (BMPs) to manage herbicide-resistant weeds are critical to the long-term sustainability of wheat production in the Pacific Northwest. Using BMPs are the most effective way to manage weeds, including herbicide-resistant weeds, especially when incorporated into a long-term weed management plan.
Start Clean!
- Plant into weed-free fields and keep them weed-free.
- Plant weed-free crop seed.
- Understand weed biology, particularly timing of seed germination, seed dormancy, and seed longevity.
- Prevent field-to-field and within-field movement by starting equipment usage in weed-free areas and by cleaning equipment after use.
- Control weeds in borders to prevent weed influx into the field.
Stay Clean!
- Scout fields routinely, and closely monitor the outcome of herbicide treatments. The sooner problems are detected, the better the chance you can adjust your management strategy.
- Use multiple herbicide mechanisms of action (MOAs) that are effective on troublesome or herbicide-resistant weeds.
- Follow the herbicide label—use the correct rate at recommended weed sizes.
- Diversify weed management practices—prevent weed seed production and reduce weed seeds in the soil seed bank.
- Use crop competitiveness and other cultural management strategies, including crop rotation.
- Use mechanical management practices, as needed.
- Manage weed seed during and after harvest to prevent weed-seed bank buildup.
- Know and understand the effects of the weed management inputs on each weed species.
Herbicide Resistant Weeds in the Pacific Northwest
- Russian Thistle
- Feral Rye
- Prickly Lettuce
- Downy Brome (Cheatgrass)
- Jointed Goatgrass
- Mayweed Chamomile
- Kochia
- Flixweed
- Tumble Mustard
- Common Lambsquarters
- Canadian Thistle (Although there are no known herbicide resistant Canada thistle in the PNW, it is a critical export contaminant.)
- Italian Ryegrass
If weeds are present after application, determine the reason! Consider the following:
- Field History—has the treatment worked before?
- Weed Biology—were weeds present at application?
- Environment—weather conditions for herbicide activity?
- Application Problems—are there clear patterns?
- Crop Cultural Practices—is the crop vigorous?
- Herbicide Resistance—seek support for suspected herbicide-resistant populations!
Seek support!
Contact your local cooperative extension office for help creating a weed management plan or if your current plan is ineffective, or see EM108: Advances in Dryland Farming in the Inland Pacific Northwest, Chapter 9, for an approach to creating such a plan.
Stay informed!
Visit the resource pages of the respective universities. UIdaho: extension.uidaho.edu/crops.aspx; OSU: agsci.oregonstate.edu/cbarc/weeds; WSU: smallgrains.wsu.edu to stay current with developments in herbicide resistance and resistance management in the region. For specific questions or concerns, contact the WSU Weed Science Team (509.335.1719; smallgrains@wsu.edu).
Acknowledgements
The authors acknowledge that many of the best management practices discussed in this publication are from the journal article titled Reducing the Risks of Herbicide Resistance: Best Management Practices and Recommendations (Norsworthy et al. 2012).
References
Corp, M., S. Machado, D. Ball, R. Smiley, S. Petrie, M. Siemens, and S. Guy. 2004. Chickpea Production Guide. Oregon State University Extension Publication EM8791-E. Oregon State University.
Evans, L.T., I.F. Wardlaw, and R.A. Fischer. 1975. Wheat. In Crop Physiology: Some Case Studies, edited by L.T. Evans, 101–150. Cambridge University Press, London.
Lyon, D.J., M.J. Walsh, J. Barroso, J.M. Campbell, and A.G. Hulting. 2019. Harvest Weed Seed Control: Applications for PNW Wheat Production Systems. Pacific Northwest Extension Publication PNW730. Washington State University.
Moss, S., L. Ulber, and I. den Hoed. 2019. A Herbicide Resistance Risk Matrix. Crop Protection 115:13–19.
Norsworthy, J.K., S.M. Ward, D.R. Shaw, R.S. Llewellyn, R.L. Nichols, T.M. Webster, K.W. Bradley, et al. 2012. Reducing the Risks of Herbicide Resistance: Best Management Practices and Recommendations. Weed Science 60(SP1):31–62.
Oelke, E.A., E.S. Oplinger, C.V. Hanson, D.W. Davis, D.H. Putnam, E.I. Fuller, and C.J. Rosen. 1991. Dry Field Pea. In Alternative Field Crops Manual. University Wisconsin Extension, Madison, WI, and University of Minnesota Extension, St. Paul, MN.
Oplinger, E.S., L.L. Hardman, E.T. Gritton, J.D. Doll, and K.A. Kelling. 1989. Canola (Rapeseed). In Alternative Field Crops Manual. University of Wisconsin Extension, Madison, WI, and University of Minnesota Extension, St. Paul, MN.
Stark, J.C. 2003. Seeding Practices. In Idaho Spring Barley Production Guide, edited by L.D. Robertson and J.C. Stark, 15–16. University of Idaho Extension Publication BUL742. University of Idaho.
Veseth, R. 1988. Crop Density—A Weed Management Tool, chap. 5, no. 11. In PNW Conservation Tillage Handbook Series. Washington State University.
By
Drew J. Lyon, Professor and Endowed Chair Small Grains Extension and Research, Weed Science, Department of Crop and Soil Sciences, Washington State University
Judit Barroso, Assistant Professor, Department of Crop and Soil Science, Oregon State University
Joan M. Campbell, Principal Researcher, Department of Plant Sciences, University of Idaho Douglas
Finkelnburg, Extension Educator, Nez Perce County Extension, University of Idaho
I.C. Burke, Professor and R. James Cook Endowed Chair, Department of Crop and Soil Sciences, Washington State University

PNW754

Published and distributed in furtherance of the Acts of Congress of May 8 and June 30, 1914, by Washington State University Extension, Oregon State University Extension Service, University of Idaho Extension, and the U.S. Department of Agriculture cooperating. WSU Extension programs, activities, materials, and policies comply with federal and state laws and regulations on nondiscrimination regarding race, sex, religion, age, color, creed, and national or ethnic origin; physical, mental, or sensory disability; marital status or sexual orientation; and status as a Vietnam-era or disabled veteran. Washington State University Extension, Oregon State University Extension Service, and University of Idaho Extension are Equal Opportunity Employers. Evidence of noncompliance may be reported through your local Extension office. Trade names have been used to simplify information; no endorsement is intended.
Pacific Northwest Extension publications contain material written and produced for public distribution. You may reprint written material, provided you do not use it to endorse a commercial product. Please reference by title and credit Pacific Northwest Extension publications.
Order Information:
WSU Extension
Fax: 509-335-3006
Toll-free phone: 800-723-1763
Email: ext.pubs@wsu.edu
OSU Extension
Fax: 541-737-0817
Toll-free phone: 800-561-6719
Email: puborders@oregonstate.edu
UI Extension
Fax: 208-885-4648
Phone: 208-885-7982
Email: calspubs@uidaho.edu
Copyright © Washington State University
Pacific Northwest Extension publications are produced cooperatively by the three Pacific Northwest land-grant universities: Washington State University, Oregon State University, and the University of Idaho. Similar crops, climate, and topography create a natural geographic unit that crosses state lines. Since 1949, the PNW program has published more than 650 titles, preventing duplication of effort, broadening the availability of faculty specialists, and substantially reducing costs for the participating states.



