
Navigating the Mite Spectrum in Honey Bee Colonies: From Harmful Tropilaelaps to Harmless Counterparts
Introduction

There are over 290 species of mites associated with honey bees (Apis mellifera Linnaeus) (Klimov et al. 2017) but only a few are considered harmful, such as tracheal mites, Varroa destructor, and Tropilaelaps spp. (Sammataro et al. 2000). Tracheal mites (Acarapis woodi), and V. destructor were introduced to the United States in the 1980s. Tracheal mites are endoparasitic mites that puncture through the walls of honey bee breathing tubes (tracheae) and feed on the hemolymph (insect “blood”). Tracheal mite infestations can lead to reduced honey bee lifespan, impaired flight ability, and colony decline or death in extreme cases. While tracheal mites are less of a concern for beekeepers now, Varroa destructor, an ectoparasitic mite, is considered a major threat to honey bee colonies due to detrimental feeding on adults and immature bees (brood) while vectoring viruses. As Varroa can rapidly reproduce inside capped honey bee brood cells, their populations peak with increased brood production. Deformed wings or bee bodies can be overt signs of damage from infestations, but other signs such as brood damage, impaired immunity, or reduced colony population can be indicative of Varroa infestations and can lead to colony collapse if left untreated.

Another parasitic mite that threatens honey bees and has not spread to North America yet belongs to the genus Tropilaelaps. Tropilaelaps spp. also feed on honey bee brood (Figure 1), causing malformation and subsequent colony decline. Varroa destructor and Tropilaelaps spp. have coexisted in Asia for decades, though reproducing in the same brood cells is less likely (Buawangpong et al. 2015). There are at least four species of mites in the genus Tropilaelaps that are known to feed on honey bee brood: T. clareae, T. koenigerum Delfinado and Baker, T. mercedesae, and T. thaii Anderson and Morgan. While all four species are closely associated with the giant honey bees (Apis dorsata and Apis laboriosa), two of these species, T. clareae and T. mercedesae, have successfully adapted to parasitizing the western honey bee, Apis mellifera, as a host (de Guzman et al. 2017; Chantawannakul et al. 2018). While Tropilaelaps causes less harm to the giant honey bees, T. mercedesae (Figure 2) is considered a major pest of western honey bees managed by beekeepers in Asia, since this honey bee species lacks evolved defenses to combat Tropilaelaps infestations.
While Tropilaelaps spp. have not been introduced to North America to date, the possible introduction would intensify colony health issues and losses that beekeepers already experience with Varroa infestations. The United States has strict regulations on the importation of honey bees, with the Honey Bee Act (7 US Code 281-286) specifically prohibiting importation of honey bee colonies, all life stages of honey bees (including germplasm), used beekeeping equipment, and untreated beeswax or honey. While enforcement of this act greatly lowers the chances of Tropilaelaps introduction, the Animal Plant Health and Inspection Service (APHIS) has been employing the annual National Honey Bee Pests and Diseases Survey (opens in new window) to surveil for Tropilaelaps in the United States since 2009. Caution must be taken when importing bees (queens and packages) in Canadian provinces to prevent inadvertent introduction of Tropilaelaps spp.
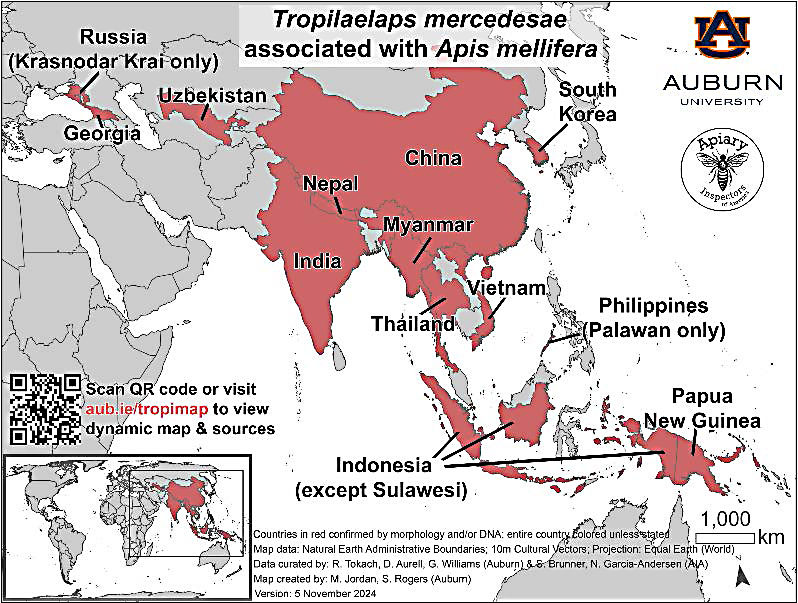
In recent years, Tropilaelaps range has expanded (Figure 3), and there is a growing concern about their potential arrival to North America (Ramsey 2021). Many other non-predatory and predatory mites are known to inhabit honey bee hives, and they mainly feed on pollen, detritus, or other mites. Beekeepers may notice these harmless mites and suspect them to be the harmful Tropilaelaps spp. This Extension publication thus aims to educate beekeepers about Tropilaelaps’ spp. current known distribution, life history, and damage to honey bee colonies, while comparing two other harmful mites, Varroa and tracheal mites. In addition, this publication will provide a short overview of examples of harmless mites that have been found through recent surveys and describe how to report suspicious mites to the Washington State Department of Agriculture (opens in new window) (WSDA) or to the Washington State University Honey Bee Program (opens in new window).
Tropilaelaps spp.
Distribution
Tropilaelaps spp. is a native pest in tropical and subtropical Asia, and their range has been expanding over the past 50 years. Their current range is not well defined but is likely to be in areas where giant honey bees (A. dorsata and A. laboriosa) are located. T. mercedesae is the species known to widely infest Apis mellifera colonies. A map from 2024 shows their present, confirmed distribution (Figure 3). The range distribution shown was collected from peer reviewed sources that used molecular or morphological techniques to document T. mercedesae associated with western honey bee colonies (Janashia et al. 2024; Brandorf et al. 2024; Mohamadzade et al. 2024). The occurrence of Tropilaelaps spp. in surrounding regions is actively being surveilled by researchers and apiary inspectors.
Biology

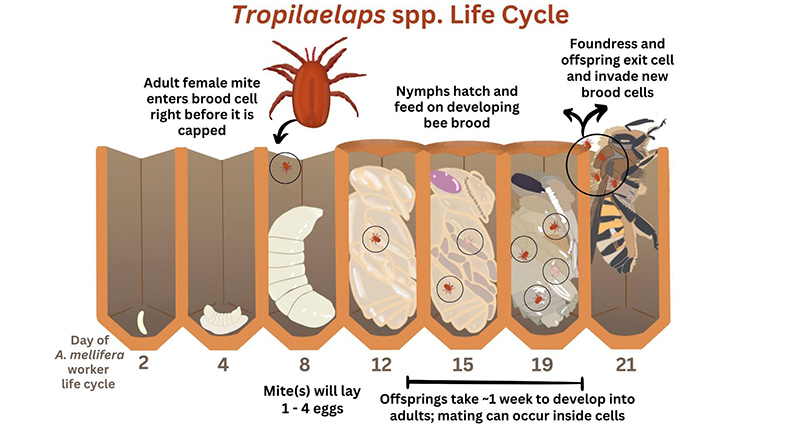
Much like Varroa, the life cycle of Tropilaelaps is dependent on honey bees. Tropilaelaps feed on honey bee brood and vector diseases, but unlike Varroa that can stay attached to honey bee adults, it is believed that the small mandibles on Tropilaelaps limit them to feeding on honey bee brood only. Therefore, Tropilaelaps requires regular access to honey bee brood to survive and reproduce; this reliance was previously thought to be a barrier to their range expansion beyond tropical areas, where honey bees can have year-round brood production. However, T. mercedesae can locally overwinter in A. mellifera colonies (Brandorf et al. 2024) and survive without brood at least three days in dry pollen and six days in empty honeycomb (Khongphinitbunjong et al. 2019). Gill et al. (2024) hypothesized that Tropilaelaps could potentially survive in a broodless colony scenario by receiving food during trophallaxis via live adult honey bees, on decaying brood, or on adult honey bees that have died during transportation. Recently, Tokach et al. (2025) observed phoresy of Tropilaelaps on honey bees exiting their colonies and suggest that these mites could spread to other locations through natural movement of A. mellifera colonies.
Tropilaelaps females are slightly larger (0.9 mm long × 0.5 mm wide) than males and are about one-third the size of Varroa (Figure 4) (de Guzman et al. 2017). Tropilaelaps are further differentiated from Varroa by their more elongated body shape (Figure 5) and their more rapid locomotion. Tropilaelaps spp. do not require a feeding period on adults like Varroa. Instead, they can emerge from one brood cell and immediately enter another cell to reproduce again. Due to a high reproductive rate, quick development time, and short time outside brood cells, Tropilaelaps populations grow very quickly.
The mature Tropilaelaps females (foundresses) lay from one to four eggs on a honey bee larva shortly before the brood cell is capped (Figure 5). Multiple foundress mites can invade a brood cell simultaneously. The mites’ offspring feed on the bee brood as they develop, and maturation is achieved in nearly one week. The average sex ratio of mite offspring is one male to every three females. The adult mites, including the foundress female(s), emerge with the adult honey bee and invade new brood cells (de Guzman et al. 2017).
Damage
Tropilaelaps spp. feed on honey bee brood and vector viruses such as deformed wing virus (Khongphinitbunjong et al. 2015). This causes stunted or deformed growth; newly emerged bees may have misshapen wings, bodies, or missing legs. Tropilaelaps spp. infestation also leads to lower bee immunity, irregular brood patterns or perforated brood cell cappings, and impaired brood production and development (de Guzman et al. 2017). In severe cases, infestations can cause the colony to collapse or honey bees to abscond from the hive.
Detecting Mites in Honey Bee Colonies
Signs of Tropilaelaps infestations can closely resemble infestation by Varroa, such as spotty brood patterns or misshapen wings. Infestation can similarly be indicative of Varroa or tracheal mite impacts, such as decreased colony performance and reduced immunity. Proper sampling methods can help beekeepers find the causative issue in their colonies (Table 1) (Pettis et al. 2013; Dietemann et al. 2013; Sammataro et al. 2013; Gill et al. 2024). Some sampling methods could be combined to improve efficacy of Tropilaelaps monitoring—for example, brood uncapping followed by the bump test (described below).
Sticky Boards
Sticky boards are typically placed under the hive on a bottom board, sticky side up, and naturally catch mites as they fall from the frames in a hive space. Natural mite drop on sticky boards can be a reliable way to detect Tropilaelaps (Gill et al. 2024). Most sticky boards have a grid printed on them to make counting mites easier. Sticky boards can be examined under a microscope or with an illuminated magnifying glass. Since Tropilaelaps and Varroa are small, this can be a time-consuming detection method, especially if you are monitoring a large number of colonies.
Bump Test
The bump test was developed by Pettis et al. (2013) and this method is currently used by APHIS as part of its annual National Honey Bee Pests and Diseases Survey. (opens in new window) It involves bumping a brood frame that contains older larvae or emerging bees onto a collection tray eight times to dislodge mites. The contents can be examined in the field with a magnifying glass or temporarily preserved in a cheesecloth and alcohol for later examination under a microscope. This method has been described as “moderately sensitive and moderately fast” for Tropilaelaps detection in the Tropilaelaps Mites 2024 United States Primer: Where We Are, What You Can Do, and Where We Are Going (links to PDF document) publication.
Examining Brood Cells
Examining brood cells or brood removal is a method used to measure and manage Varroa or Tropilaelaps infestations in honey bee colonies. This method can involve randomly removing 100–200 capped brood from the frame or removing large portions with an uncapping fork to observe the number of brood infested. Mite populations can be estimated by counting the number of mites found in the brood cells. Using an uncapping fork to remove small, sporadic areas of capped drone or worker brood has been effective for measuring Varroa mite levels (Dietemann et al. 2013).
For Tropilaelaps detection, Gill et al. (2024) found that uncapping infested brood with tweezers was more effective than using an uncapping fork, as published in their study. However, during certain stages of brood development, the mites tended to stay within the cell and could be dispersed by blowing over the brood or using a paintbrush to “sweep the cell.” Additionally, Uzunov et al. (2024) published a novel method, “rapid brood decapping,” which uses cosmetic wax strips and 60 seconds of videotaping to document emerging mites. They reported that this method improved ease of sampling and time efficiency for beekeepers and produced results comparable to the traditional brood cell examination method by Dietemann et al. (2013), with approximately 90% similarity.
Table 1. Suitable sampling methods for harmful mites in honey bee colonies.| Diagnostic Test | Tropilaelaps Mites | Varroa Mites | Tracheal Mites |
|---|---|---|---|
| Sticky Boards | *** | *** | X |
| Bump Test | *** | *** | X |
| Examining Brood Cells (removal or uncapping) | *** | *** | X |
| Alcohol Wash | X | *** | X |
| Powdered Sugar Shake | * | * | X |
| Microscopic Dissection | X | X | *** |
Sources: Extrapolated from work by Pettis et al. (2013), Dietemann et al. (2013), Sammataro et al. (2013), and Gill et al. (2024).
Notes: *** = most suitable, * = suitable, X = least suitable or not applicable.
Alcohol Wash
Alcohol washes are an extremely effective way to monitor for Varroa (Dietemann et al. 2013) since the mites cling to adult honey bees and feed on their fat body and hemolymph (insect blood). Although, for Tropilaelaps, it may not be the best option since Tropilaelaps do not stay on honey bee adults. This method is most appropriate for detecting Varroa. To perform an alcohol wash, honey bees are collected from brood frames with older larvae or emerging bees, placed into a jar containing 70% alcohol, and shaken for one minute. The honey bees and wash solution are poured through a mesh screen into a collection bin. Any mites transferred to the collection bin can be counted or observed.
Powdered Sugar Shake
Powdered sugar shakes are another popular monitoring method for Varroa since the mites cling to adult honey bees. Although, for Tropilaelaps, it may not be the best option since Tropilaelaps do not stay on honey bee adults. Like the alcohol wash, this method is most appropriate for detecting Varroa. In this method, honey bees are collected from brood frames with older larvae or emerging bees, placed into a jar with a screened lid, and gently rolled around for one minute. The powdered sugar dislodges mites from the bees and mites can be shaken from the jar into a collection bin. Using a spray bottle, the contents in the collection bin can be sprayed to dissolve the powdered sugar, which makes it easier to find any dislodged mites, then the number of mites can be counted or observed.
Microscopic Dissection
Since tracheal mites live inside the honey bees’ tracheal system, they are difficult to detect externally; therefore, microscopic techniques such as dissecting honey bees is the ideal diagnostic method for tracheal mite detection (Sammataro et al. 2013). Tracheal mites are more attracted to young honey bees, so honey bees are collected from brood frames and dissected to examine their tracheal system for the presence of mites. Typically, at least 20 bees are dissected to determine presence or absence of tracheal mites at the colony level. This method requires some training to avoid damaging the trachea and to accurately identify the mites and is not recommended for detecting Tropilaelaps or Varroa mites.
Management
The APHIS has been employing the annual National Honey Bee Pests and Diseases Survey (opens in new window) to surveil for Tropilaelaps in the United States since 2009. Each year, beekeepers with at least eight colonies in their apiary can participate for free, contribute to national surveillance of any exotic pests like Tropilaelaps mites, and receive information about their apiary’s health, including the presence or absence of common honey bee viruses as well as populations of Varroa and Nosema, a common microsporidian parasite.
If you would like to volunteer to participate in the National Honey Bee Pests and Diseases Survey for Washington State, please contact the WSU Bee Program at entomology.bees@wsu.edu (opens new email message)
Management efforts are currently being researched in regions where Tropilaelaps is already established. The United States Department of Agriculture (USDA) and Environmental Protection Agency (EPA) assert that existing registered chemicals for Varroa control may be effective options for Tropilaelaps control. As stated in the Tropilaelaps Mites 2024 United States Primer: Where We Are, What You Can Do, and Where We Are Going (links to PDF document): “Under FIFRA Section 2(ee), a federal statute for EPA regulation of pesticides, it is not required to have Tropilaelaps, specifically T. mercedesae, listed as a specific mite on the label in the United States. Any miticide products that are used properly in accordance with label directions (i.e. are labeled for use in bee colonies to kill mites), restrictions, and application parameters, could be used to control Tropilaelaps.” Note that while at the federal level, FIFRA 2(ee) ensures the legal use of registered products that list honey bee colonies as a use site, regardless of target pest, state regulations may be more restrictive.
A current list of federally registered pesticide products for Varroa control that include formic acid, oxalic acid, hops beta acids, amitraz, tau-fluvalinate, and coumaphos can be found at the EPA website (opens in new window). Thus far, research has shown formic acid and brood breaks to be the most promising methods, but research is still being conducted to determine the efficacy of Varroa control products for efficacious Tropilaelaps control (Tokach et al. 2024). Additionally, since Tropilaelaps likely do not feed on adults, using cultural control methods, such as brood breaks or removing frames of developing bees from a colony, will likely be a powerful control tool in reducing Tropilaelaps infestations. Canada’s Pest Management Regulatory Agency (PMRA) may have additional guidelines on efficacy standards and registered use of different products.
Other Mites Associated with Honey Bee Colonies

Honey bee nests are suitable habitats for nonparasitic, omnivorous, or pollen-feeding mite species. Many live on the hive floor, feeding on bee debris or fungi (Sammataro et al. 2000). Given the high diversity of bee-associated mites, some may resemble Tropilaelaps. With increased vigilance about potential Tropilaelaps introduction to North America, beekeepers may notice mites inhabiting the bottom boards or hive crevices and become concerned. While beekeepers are always encouraged to report sightings of suspicious mites, this section aims to inform beekeepers about the potential mite diversity within a honey bee colony.
One example is the pollen mite (Mellitiphis alvearius), which is light brown and similar in size to Tropilaelaps (Figure 6), but pollen mites are not destructive to honey bees and do not impact colony health. Another example of a look-alike mite is the house fly mite (Macrocheles muscaedomesticae; Figure 7). This species has been long considered harmless to honey bee colonies, but a recent case of high-density phoresy on A. mellifera was observed to detrimentally impact the honey bees’ ability to fly in Texas, USA (Fulton et al. 2025).

Some samples from the APHIS National Honey Bee Pests and Diseases Survey have contained harmless mites collected by bump tests in the Pacific Northwest region of the United States. In 2023, a suspicious mite was flagged, but it was identified as Hypoaspis lubrica (Figure 8), a soil-dwelling predatory mite that feeds on other mites and can be found in bee colonies when there are certain conditions such as excess hive debris, tall grass, or poor ventilation. Other mites passively captured in the bump tests were identified as Macrocheles sp. (Figure 9), Neocypholaelaps sp. (Figure 10), or Parasitellus sp. (Figure 11). Macrocheles sp. live in a variety of habitats, including manure, dung, litter, decaying organic substances, and nests of social bees, where they feed on small invertebrates. Neocypholaelaps sp. (Figure 10) and Parasitellus sp. (Figure 11) normally live on flowers of various plants and feed on pollen and nectar, and can feed on provisioned pollen in honey bee nests.




Diseases Survey bump test sample contents from the Pacific Northwest. Photo courtesy: Kensie Olson, University of Maryland Bee Lab.
Reporting Suspicious Mites
If you think you may have Tropilaelaps in your hive(s) or see any suspicious mites, report them to the Washington State Department of Agriculture’s (WSDA) Pollinator Program immediately. To report a possible sighting of Tropilaelaps, please visit the Report Invasive Pest Sightings (opens in new window) webpage in addition to contacting the WSDA Pollinator Health Coordinator (opens in new window) and WSU Bee Program (opens in new window).
Submit a photo of the suspected mite along with the report, and if possible, collect specimen(s) in alcohol by gently picking up the mite(s) with a paintbrush and submerging the brush in alcohol to dislodge the mite(s). A site visit by WSDA or WSU Bee Program staff will likely occur for sample collection; potential Tropilaelaps sightings will be taken very seriously. Confirmation of a positive Tropilaelaps mite infestation will involve a response from the USDA.
The response to a positive Tropilaelaps mite infestation will vary depending on how widespread the infestation is upon investigation. In the past, attempts to control the spread of Varroa, when it was first introduced, involved quarantines and hive destructions; this proved to be unsuccessful. It’s more likely that the response to a positive Tropilaelaps infestation will involve an immediate and widespread education and outreach campaign on identification and control options. However, the potential arrival of this pest to North America will be detrimental to the beekeeping and agricultural industries in the United States, and exhaustive efforts will be made to prevent Tropilaelaps from becoming established in North America.
WSDA Contact Information
Dr. Katie Buckley, Pollinator Health Coordinator, kbuckley@agr.wa.gov (opens new email message), 360-480-0602.
WSU Bee Program Contact Information
entomology.bees@extension-pubs (opens new email message)
Other Useful Resources About Tropilaelaps spp.
- Tropilaelaps fact sheet—Tropilaelaps A Growing Threat to Honey Bees What You Need to Know (links to PDF document) by the Apiary Inspectors of America, Auburn University, Honey Bee Health Coalition, Pollinator Partnership, Project Apis m., US Department of Agriculture Animal and Plant Health Inspection Service and US Department of Agriculture Agricultural Research Service
- Tropilaelaps Training 2024 Documentary Series and 2024 Tropilaelaps Training in Thailand Trailer (opens in new window)—Project Apis m.
- Tropi STOP webpage (opens in new window)—Pollinator Partnership and partners
- Tropilaelaps Mites (opens in new window)—Honey Bee Pests from Apiary Inspectors of America
- Tropilaelaps: What Beekeepers Need to Know (opens in new window)—webinar recording with Dr. Samuel Ramsey
- Tropilaelaps Infestation of Honey Bees (opens in new window)—case definition publication by US Department of Agriculture
- Featured Creatures Collection: Tropilaelaps mite—publication from University of Florida
References
Brandorf, A., M.M. Ivoilova, O. Yañez, P. Neumann, and V. Soroker. 2024. First Report of Established Mite Populations, Tropilaelaps mercedesae, in Europe (opens in new window). Journal of Apicultural Research 1–3.
Buawangpong, N., L. De Guzman, K. Khongphinitbunjong, A. Frake, M. Burgett, and P. Chantawannakul. 2015. Prevalence and Reproduction of Tropilaelaps mercedesae and Varroa destructor in Concurrently Infested Apis mellifera Colonies. Apidologie 46.
Chantawannakul, P., S. Ramsey, D. vanEngelsdorp, K. Khongphinitbunjong, and P. Phokasem. 2018. Tropilaelaps Mite: An Emerging Threat to European Honey Bee. Current Opinion in Insect Science 26: 69–75.
de Guzman, L.I., G.R Williams, K. Khongphinitbunjong, and P. Chantawannakul. 2017. Ecology, Life History, and Management of Tropilaelaps Mites (opens in new window). Journal of Economic Entomology 110(2): 319–332.
Dietemann, V., F. Nazzi, S.J. Martin, et al. 2013. Standard Methods for Varroa Research (opens in new window). Journal of Apicultural Research 52(1): 1–54.
Fulton, J.C., S.J. Bolton, M.R. Moore, S. Kyles, P. Klimov, and J.D. Ellis. 2025. Macrocheles muscaedomesticae, a First Record of Phoresy on Apis mellifera. Journal of Apicultural Research 1–3.
Gill, M.C., B. Chuttong, P. Davies, et al. 2024. Assessment of the Efficacy of Field and Laboratory Methods for the Detection of Tropilaelaps spp (opens in new window). PLoS ONE 19(9): e0301880.
Janashia, I., A. Uzunov, C. Chen, C. Costa, and G. Cilia. 2024. First Report on Tropilaelaps mercedesae Presence in Georgia: The Mite Is Heading Westward! (opens in new window) Journal of Apicultural Science 68(2).
Khongphinitbunjong, K., L.I. de Guzman, M.R. Tarver, T.E. Rinderer, and P. Chantawannakul. 2015. Interactions of Tropilaelaps mercedesae (opens in new window), Honey Bee Viruses and Immune Response in Apis mellifera. Journal of Apicultural Research 54(1): 40–47.
Khongphinitbunjong, K., P. Chantawannakul, O. Yañez, and P. Neumann. 2019. Survival of Ectoparasitic Mites Tropilaelaps mercedesae in Association with Honey Bee Hive Products (opens in new window). Insects 10(2): 36.
Klimov, P.B., B.M. OConnor, R. Ochoa, G.R. Bauchan, and J. Scher. 2017. Bee Mite ID—an Online Resource on Identification of Mites Associated with Bees of the World (opens in new window). Journal of Acarological Society of Japan 26(1): 25–29.
Mohamadzade, N., O. Joharchi, S. Aryal, et al. 2024. Exploring Genetic Variation and Phylogenetic Patterns of Tropilaelaps mercedesae (Mesostigmata: Laelapidae) Populations in Asia (opens in new window). Frontiers in Ecology and Evolution 21(12).
Pettis, J.S., R. Rose, E.M. Lichtenberg, et al. 2013. A Rapid Survey Technique for Tropilaelaps Mite (Mesostigmata: Laelapidae) Detection (opens in new window). Journal of Economic Entomology 106(4): 1535–1544.
Ramsey, S.D. 2021. Foreign Pests as Potential Threats to North American Apiculture: Tropilaelaps mercedesae, Euvarroa spp, Vespa mandarinia, and Vespa velutina (opens in new window). Veterinary Clinics: Food Animal Practice 37(3): 545–558.
Sammataro, D., U. Gerson, and G. Needham. 2000. Parasitic Mites of Honey Bees: Life History, Implications, and Impact (opens in new window). Annual Review of Entomology 45: 519–548.
Sammataro, D., L. de Guzman, S. George, R. Ochoa, and G. Otis. 2013. Standard Methods for Tracheal Mite Research (opens in new window). Journal of Apicultural Research 52(4): 1–20.
Tokach, R., B. Chuttong, D. Aurell, L. Panyaraksa, and G.R. Williams. 2024. Managing the Parasitic Honey Bee Mite Tropilaelaps mercedesae Through Combined Cultural and Chemical Control Methods (opens in new window). Scientific Reports 14: 25677.
Tokach, R., D. Aurell, B. Chuttong, and G.R. Williams. 2025. Observation of Tropilaelaps mercedesae (Mesostigmata: Laelapidae) on Western Honey Bees (Apis mellifera) Exiting Colonies. Journal of Economic Entomology 118(2): 966–969.
Uzunov, A., I. Janashia, C. Chen, C. Costa, and M. Kovačić. 2024. A Scientific Note on “Rapid Brood Decapping”—a Method for Assessment of Honey Bee (Apis mellifera) Brood Infestation with Tropilaelaps mercedesae (opens in new window). bioRxiv.
Authors:
Briana E. Price, Extension Coordinator,
Department of Entomology, Washington State University
Katie Buckley, Pollinator Health Coordinator,
Washington State Department of Agriculture
Brandon K. Hopkins, Assistant Professor,
Department of Entomology, Washington State University
Priyadarshini Chakrabarti, Assistant Professor,
Department of Entomology, Washington State University

FS399E





