Gardening with Companion Plants (Home Garden Series)
Popularized Forms of Companion Planting
Gardeners often hear about “companion planting” as they prepare their vegetable gardens and flower beds. Popular books, charts, and online resources about companion plants are readily available, but most lack any credible, supporting science.
- Folklore companion plants (Figure 1A). Many centuries ago, plants were associated with the four elements (earth, air, fire, and water) and signs of the zodiac. One of the outcomes of this belief system is the practice of planting together those species that “love” each other (Jeavons 2012; Riotte 2004), and “snuggle for protection” (Hemenway 2001). Assigning human characteristics and emotions to other species, which is called anthropomorphism, allows people to feel more connected to the rest of the living world.
- Companion plants determined through sensitive crystallization (Figure 1B). This is one of several pseudoscientific practices included in biodynamics, which is a form of organic agriculture developed almost a century ago (Chalker-Scott 2013). Sensitive crystallization involves interpreting patterns that plant extracts make as they dry, allowing practitioners to declare which plants are compatible with one another (Philbrick and Gregg 1966; Riotte 2004).
- Permaculture companion plants (Figure 1C). Permaculture is a philosophical belief system that includes both scientific and pseudoscientific practices. Practitioners believe that plants do not necessarily compete with one another (Holmgren 2002), so lists of recommended plants include invasive species (Mollison 1994). For example, companion planting includes the use of Scotch broom (a listed noxious weed in some states, including Washington; https://www.nwcb.wa.gov/weeds/scotch-broom) and other related broom species as nitrogen-fixing companion plantings for fruit trees. Not only does the broom compete for water and nutrients but it produces vast numbers of seed that only add to the spread of this noxious weed.
Popularized versions of companion planting are complicated, contradictory, and confusing. The science behind companion planting is substantial, enlightening, and more practical than any myth-based information.


Science-Based Companion Planting
From a scientific perspective, companion planting unites two fields of study: agriculture and ecology. Agricultural research has provided us with the practices of polyculture and intercropping, which involve planting mutually beneficial species. Ecological studies have described naturally occurring, beneficial associations among plants in nonagricultural environments. Together, these two sciences define symbiotic relationships among plants and their associated, beneficial insects and microbes.
In the scientific literature you will find a variety of phrases used interchangeably with companion planting. They include:
- Intercropping—Generally used to describe relatively simple systems, where one or a few species are used in alternate rows; agricultural terminology (Figure 2A).
- Plant association—Plant species that are often found together in natural environments; ecological terminology (Figure 2B).
- Nurse plant (or pioneer species)—A plant that enhances survival of newly germinated seedlings by providing shade, blocking wind, or otherwise moderating the microclimate; ecological terminology (Figure 2C).
- Polyculture—More complex than intercropping, as it involves multiple species; often used to describe hedgerows, windbreaks, and other multispecies plant systems; agricultural and ecological terminology (Figure 2D).




Whenever a plant begins to grow in the garden, it alters its environment physically, chemically, and biologically (Table 1). Some of these changes—reduced soil water, nutrients, and sunlight, for instance—make it more difficult for neighboring plants to grow. But other alterations—increased shade, decreased evaporation and wind—may enhance the germination, growth, and survival of some species. In ecology, pioneering species are those that pave the way for plants less adaptable to extreme environmental conditions (Close et al. 2005). Pioneering species may be nitrogen-fixers (like beans, peas, and other legumes), shade providers, or serve as habitats formicrobes and insects. Complex associations of plants, insects, and microbes can have positive, negative, or minimal effects on one another. When we are able to tease out the positive associations, we can use the information for planting and maintaining our gardens.
All of the benefits of companion plants described in Tables 1A-C have been documented through research and used in the management of agricultural, ornamental, and restoration landscapes.
Table 1A. Physical modifications made by plants.
| Physical alterations | Examples |
|---|---|
| Compaction reduction | All plants will reduce soil compaction, through insulation above or root support below |
| Wind block | Taller shrubs and trees can redirect wind |
| Shade | The amount of shade provided depends on plant size, persistence of leaves, and other morphological characteristics |
| Temperature moderation | Low-growing species, such as ground covers, protect soil from temperature fluctuations |
| Moisture conservation | Increased shade and temperature moderation help conserve soil moisture |
| Structural support | Sturdy, upright species provide vertical space for climbing plants |
| Erosion control | Roots of all plants help reduce erosion from water runoff; low growing species, such as ground covers, reduce erosion due to wind |
Table 1B. Chemical modifications made by plants.
| Chemical alterations | Examples |
|---|---|
| Increased organic matter | All plant material left on soil will be naturally incorporated into the soil profile |
| Nitrogen fixation | Nitrogen-fixing species including legumes, alder, and hundreds of other species* |
| Nutrient availability | Increasing densities of plants will decrease nutrient levels |
| Water availability | Increasing densities of plants will decrease water availability |
| Salt accumulation | Salsola soda, Portulaca oleracea, and other species found in high-salt environments take up salt, allowing more sensitive plants to grow* |
| Heavy metal accumulation | A variety of plants can take up and retain heavy metals* |
| Volatile organic compounds (VOCs) | Many plants can produce VOCs which can have attractant or repellent effects on other species* |
| *See additional information in text. |
Table 1C. Biological modifications made by plants.
| Biological alterations | Examples |
|---|---|
| Weed control | Dense plantings can reduce or eliminate weeds (Breitenmoser et al. 2022) |
| Pest control | A diverse planting palette can interfere with pest feeding and reproduction, and provide habitat for natural predators of pest insects* |
| Disease control | Plant diversity decreases likelihood of disease spread, as many pathogens are host specific* |
| Mycorrhizal enhancement | Many desirable plant species, especially woody species, are colonized by beneficial mycorrhizal fungi* |
| *See additional information in text. |
Chemical Alterations
1. Nitrogen fixation
While gardeners know about the value of nitrogen-fixing vegetables like peas and beans, they are often unaware that many other species besides legumes can add nitrogen to the soil (Tedersoo et al. 2018). Nitrogen-fixing trees, shrubs, perennials, and annuals (Figures 3A–D) can transfer nitrogen to the soil microbiome in a process called rhizodeposition (Fustec et al. 2010). Some nitrogen-fixers pull double duty, both increasing the nitrogen levels of the soil as well as attracting pollinators (Abad et al. 2019).
Though this process does not directly supply other plants with nutrients, many of the beneficial bacterial and fungal species that are recipients also colonize and benefit plant roots. There is also some evidence that fine roots of neighboring plants can take up nitrogen as well if they are in the right place at the right time (Paula et al. 2015). Nitrogen-fixing species enrich the soil by improving its microbiome; they do not deliberately provide nitrogen to other plant species nor do nitrogen-fixing companion plants always improve the health and survival of other plants (Milne et al. 2021). Furthermore, yields of the nitrogen-fixing species can suffer if they are not competitive with the other crop plants (Monti et al. 2016).




2. Salt and heavy metal accumulation
Salty garden soils, such as those found in arid or marine environments, can benefit from the addition of salt-tolerant species. Strawberries (Karakas et al. 2021), peppers (Colla et al. 2006), tomatoes (Albaho and Green 2000; Graifenberg et al. 2003; Green et al. 2000; Karakas et al. 2016), and other vegetables grown in moderately salty soils may do better when grown with salt-tolerant companion plants such as Portulaca oleracea, Salsola soda, and Suaeda salsa (Figure 4). These plants take up both sodium and chloride, reducing the impact to less tolerant species.
Just as plants adapted to saline soils can be used to remove dissolved salts, plants adapted to high levels of toxic heavy metals (Figure 5) can detoxify, or phytoremediate, soil contaminated with such chemicals. Such species have been of great interest to scientists (Rascio and Navari-Izzo 2011), who suggest that plants hyperaccumulate heavy metals as a defense mechanism against natural enemies, such as herbivores. While little research has been published with direct relevance to gardens and landscapes, we generally understand that many wetland species, such as Salix and Populus, are phytoremediators.


3. Volatile organic compounds (VOCs)
Many plants release aromatic chemicals, also known as volatile organic compounds (VOCs) and “essential oils,” that are intercepted by other plants or animals (Figure 6). Some animals, including pollinators, cats, and humans, find the scents of certain mints attractive; others are repelled by the odors. Researchers have identified many of these compounds and found them to be repellent to insect pests in laboratory experiments (Dieudonné et al. 2022; Pouët et al. 2021), decreasing feeding behavior and ultimately the insects’ ability to reproduce and survive (Ameline et al. 2022; Borges et al. 2022; Dardouri et al. 2021).
Pest-repelling properties of mints and other aromatic species have long been a staple of popular companion planting wisdom, but field research has not borne out this belief for mint or any other aromatic species (Finch et al. 2003; Laffon et al. 2022). The failure of lab success to translate to practical application is not surprising: VOCs in the atmosphere dissipate quickly with increased distance from the plant that releases them. Aromatic plants in closed systems such as greenhouses (where VOCs could accumulate) might be useful as pest repellents (Nyalala and Grout 2007), but gardens and landscapes are unlikely to be affected.
VOCs are also discussed later in this publication in context of how they can influence the presence of beneficial and pest insect species.



Biological Alterations
1. Enhance biodiversity
The biggest change that additional plant species bring to a garden or landscape is an immediate increase in plant diversity. While the addition of only one companion plant may not have a positive outcome in reducing a particular pest, multiple companion species increase the odds of successful pest control (Sutter et al. 2022). Increased plant diversity attracts more herbivores, which in turn attract other species that eat or parasitize them: this creates a more complex food web. A complex food web is more resilient than a simple one (Figure 7); it can more easily tolerate and recover from disruptions.
When more plant species are added to a system, the system becomes more biodiverse by providing new habitat and resources for insects, birds, reptiles, mammals, and other plants (Chalker-Scott 2018, 2015a). The other biological alterations that companion plants create are all related to improved biodiversity.
2. Attract and support beneficial insects and birds
- Provide alternative food and shelter for generalist predator insects and parasitoids (Balzan 2017; Hatt et al. 2019; Saldanha et al. 2019; Wang et al. 2020). These “insectary plants” support beneficial insects such as hoverflies (Gospodarek 2021), lady beetles (Gospodarek2021; Wang et al. 2020), and predatory bugs (Chailleux et al. 2022; Zuma et al. 2022).
- Improve yield and quality of crops such as strawberry by attracting additional pollinators (Griffiths-Lee et al. 2020).
- Provide habitat for predacious insects (Gontijo et al. 2018) and insectivorous birds through addition of vertically structured landscapes (Figure 8) (Chalker-Scott 2015a).

3. Reduce plant damage by repelling or disrupting the behavior of harmful insects
Probably the best-known example of repellent companion planting is the use of French marigolds (Tagetes patula) in annual beds and vegetable gardens. While this species has been found to have a negative effect on pests such as codling moths, it also discourages the moths’ natural enemies and other beneficial insects (Laffon et al. 2022). These marigolds do not distinguish between pest and beneficial insect species.
To improve our understanding of repellent species, researchers have proposed the “disruptive crop hypothesis” which states that the presence of nonhost plants in a given area disrupts the ability of specialist herbivorous insects to discover their appropriate host plants for feeding or egg-laying (Mansion-Vaquie et al. 2020). Insects spend more time searching for their host, and as a result, spend less time on host plants overall when their search is disrupted, all the while consuming valuable metabolic resources without feeding or reproducing.
The disruptive crop hypothesis seems to support the idea that VOCs can attract or repel pests—but field research indicates that neither aroma nor taste influences the ability of herbivorous insects to find their host (Finch and Collier 2012; Finch et al. 2003). Instead, it is the presence of green objects—nonhost leaves or otherwise—that disrupts host searching (Figure 9). Insects seek out green surfaces on which to land and at that point rely on the smell of plant chemicals to verify their choice (Finch and Collier 2012, 2000; Nottingham 1988).
A concept called “push-pull intercropping” suggests that crops can be interplanted such that one nonhost species repels (or pushes) the pest insect while another attracts (or pulls) the pest, leaving the host species less damaged. However, insects are not repelled from landing on nonhost plants nor do VOC-emitting “trap crops” attract insects more than five meters away (Finch and Collier 2012). Instead, research suggests that the number of green objects surrounding the host plants is the major factor that prevents insect pests from finding their host plants (Collier and Finch 2003). The visual miscues provided by nonhost plants force insects to choose between appropriate (host) and inappropriate (nonhost) leaves on which to land: for intercropping to be effective, insects must land on the nonhost plants (Finch and Collier 2012). Thus, perimeter plantings of nonhost plants around crops are less effective with increased distance from the perimeter (Conboy et al. 2019), but intercropped rows can be effective (George et al. 2019).
Many pest insects not only damage crops directly but carry bacterial, fungal, and viral diseases. Intercropping susceptible crop plants with nonhost plants has been found to reduce the spread of aphid-borne viruses (Waweru et al. 2021). Similarly, organisms associated with protecting pest insects can be lured away from the host species of concern, leaving the pests open to predators and parasitoids. An example of this was seen with apple aphids, who are protected by black garden ants that eat the honeydew. When bean plants were planted within the orchard, the ants preferred to visit the bean aphids; apple aphid populations were nearly eradicated (Pålsson et al. 2020).
Increased biodiversity through companion planting can lead to unexpected and unwanted results. For instance, providing additional food resources for lady beetles can reduce their consumption of aphids (Wang et al. 2020). Likewise, populations of beneficial parasitoids can be reduced by companion plants that attract predators of the parasitoids (Saldanha et al. 2019). Enhanced structural diversity of vegetation may, in some cases, interfere with the ability of nonflying predators to reach their prey (Gontijo et al. 2018). These drawbacks are minimal, however, compared to the significant reductions in many pest species when gardens and landscapes are home to a wide diversity of annual, perennial, and woody plants.
4. Provide mycorrhizal inoculants and their associated benefits
Increasing research on belowground plant relationships has revealed that many plants share indirect connections among their roots, primarily through mycorrhizal relationships (Figure 10). The benefits of mycorrhizae are well known (Chalker-Scott 2017), and they can colonize woody plants, perennials, grasses, and even annuals. Once established in a root system, mycorrhizae provide enhanced uptake of water and nutrients, and in doing so can improve plant resistance to drought stress and diseases. The importance of mycorrhizal interconnections in establishing new plants has long been recognized in ecological restoration research (McGee 1990). However, many crop plants have been regarded as nonmycorrhizal until recently.
Conventional agriculture is still dominated by monocultural crops planted in annually tilled soils. These conditions are not ideal for mycorrhizae (Chalker-Scott 2017), so it is not surprising that they are rarely associated with annual crop plants. When “nonmycorrhizal” crops are intercropped with other species, they can become mycorrhizal (Kellogg et al. 2021). This phenomenon may help account for reports that intercropped species may be more productive in the presence of the companion plant (Hata et al. 2019) rather than suffering from increased competition for underground resources.

Using Evidence-Based Companion Planting Strategies in Home Gardens and Landscapes
- When creating buffer strips, hedgerows, or other plantings that will act as refuges for beneficial wildlife, select a wide variety of species. Limiting companion plants to just a few species can result in little to no biological control activity (Lérault et al. 2021; Lopez and Liburd 2022).
- Be aware that perennial companion plants will take a year or two to establish. Thus, their optimum benefits may not be realized until that time (Montoya et al. 2020). Annual companion plants should be used if immediate benefits are desired.
- If you are growing perennial crops, avoid using annual companion plants that require yearly soil disruption. Crop growth and yield can be negatively affected (LaMondia et al. 2002).
- Dense ground covers function as living mulches and will reduce weeds (Breitenmoser et al. 2022). To reduce possible competition with other plants, use living mulches on pathways, between rows in vegetable gardens and orchards, and other locations that are not densely planted. Living mulches play a crucial role in protecting soil from erosion as well as biological and chemical degradation, and this improvement may outweigh any drawbacks from competition (Adamczewska-Sowińska et al. 2022).
- To reduce competition among desirable plants, choose species whose roots are less likely to interfere with one another. Intersperse large taproot vegetables like carrots and radishes with those whose root systems are shallow and widespread, like corn, onions, and lettuces.
- To reduce possible predation among beneficial insects (such as lady beetles), use a wide variety of companion plants that will provide floral resources as a supplemental food source for predacious insects (Liang et al. 2022).
- Do not use invasive species or even aggressive native plants. Some companion plants can be overly competitive for other resources like sunlight, resulting in reduced growth and vigor of other species (Urli et al. 2020).
- Use nitrogen-fixing species to improve overall soil microbiome health and reduce the need for nitrogen fertilizers (Akinnifesi et al. 2010).
- Experiment with different densities of species to determine satisfactory yields from each (Gospodarek 2021).
- Be cautious when using strong water sprays on plants, as this can dislodge predatory insects as well as their prey (Chailleux et al. 2022).
- Avoid using broad-based insecticides, as they will kill pest predators and parasitoids along with the pest. Proper companion planting will reduce the need for pesticides in general (Balmer et al. 2013).
- A well-chosen organic mulch can improve plant growth and productivity better than the use of nitrogen-fixing companion plants (Milne et al. 2021). A combination of organic mulch and companion plants has also been found to be more effective than companion planting alone (Waiganjo et al. 2007).
- Use a woody organic mulch, such as arborist wood chips, to enhance mycorrhizal populations, improve overall soil health, and control weeds. Arborist wood chip mulches also house predatory spiders and insects, such as ground beetles (Chalker-Scott 2015b).
- In vegetable gardens, try to intercrop different species so that individuals of the same species are as far apart as possible from each other. This will reduce the ability of pest insects to infest an entire crop.
- Consider using green cards or other non-plant surfaces to further confuse herbivorous insects (Martins et al. 2009). Be aware this will also affect some desirable species, such as butterflies and moths whose larval life cycle is spent on host plants.
- Use diverse flowering species around greenhouses to enhance beneficial species and reduce pest abundance. This will reduce the likelihood of pests entering the greenhouse accidentally and reduce the need for insecticide use in the greenhouse (Li et al. 2020).
- Instead of using wooden planting structures for viny vegetable and fruit support, consider using live stakes instead. To keep the stakes at a manageable size, prune them as needed but leave the prunings as a mulch. Not only will live stakes enhance your garden’s biodiversity, but the benefits of the additional mulch can result in substantial increases in crop yield (Otu and Agboola 1994).
- Consider experimenting with companion plants that have been identified as having natural insecticidal properties on specific pests but whose effects on beneficial insects are unknown (Amoabeng et al. 2019). Do you notice an increase or decrease in the presence of beneficial insects as well as pest species? Plants that have negative effects on beneficial species would not be good choices for companion planting (as we have discovered with Tagetes patula earlier [Laffon et al. 2022]), but a plant that discourages pests without impacting beneficials would be desirable as a companion plant.
The Three Sisters
One of the best-known examples of companion planting is the combination of corn, beans, and squash (often called the “Three Sisters”). These vegetables provide three different, important functions to their assemblage: beans are legumes that fix nitrogen, corn stalks provide physical structure for the beans to twine around, and squash vines carpet the soil as a living mulch, keeping the soil cool, moist, and shaded, while suppressing weed growth. Popular books make claims, without evidence, that “The Three Sisters produce more food, with less water and fertilizer, than a similar area planted to any one of these three crops in isolation” (Hemenway 2001). It is not surprising that the appeal of the Three Sisters and other companion planting combinations remains high among gardeners.

There are few peer-reviewed publications on the practical benefits of the Three Sisters methodology. Some articles discuss the history of the practice (Mt. Pleasant and Burt 2010; Ngapo et al. 2021) and the importance of these crops to Indigenous peoples (Kapayou et al. 2023). Those that contain experimental field data comparing monocultures versus polycultures of these three crops report:
- No significant differences in plant nutrient content (Kapayou et al. 2023).
- No significant differences in soil characteristics (Kapayou et al. 2023).
- Reduced yields of beans and squash in polyculture (Mt. Pleasant and Burt 2010).
- Increased productivity in polyculture using land equivalent ratios (LER) (Mt. Pleasant and Burt 2010; Zhang et al. 2014).
This last outcome has been used to promote the Three Sisters as a superior method of producing corn, beans, and squash together. The figure below explains LER graphically.
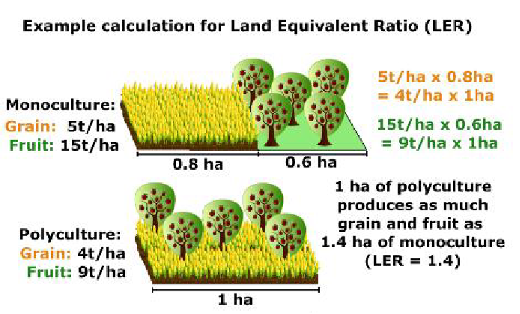
“The land equivalent ratio is a concept in agriculture that describes the relative land area required under sole cropping (monoculture) to produce the same yield as under intercropping (polyculture)” (Wikipedia 2022).
While this model is mathematically accurate, like any model it will be altered by local conditions. The yield of the Three Sisters crops will be different in the hot and dry Southwest than in the relatively cooler and wetter Northeast. Productivity of each crop would need to be found before LER could be determined for any locality. Furthermore, the LER does not take into account the costs and benefits associated with different cropping systems (Khanal et al. 2021). It may be a useful model for farmers with large acreage, but has little practical value in a home garden or other limited space.
Though there may be historic or cultural reasons for planting a Three Sisters polyculture vegetable garden, there is no compelling, published evidence that it benefits plant productivity or soil quality.
References
Abad, M.K.R., S.A.A. Fathi, G. Nouri-Ganbalani, and B. Amiri-Besheli. 2019. Influence of Tomato/Clover Intercropping on the Control of Helicoverpa armigera (Hübner). International Journal of Tropical Insect Science 40(1): 39–48. DOI: http://dx.doi.org/10.1007/s42690-019-00048-z.
Adamczewska-Sowińska, K., W. Wojciechowski, M. Krygier, and J. Sowiński. 2022. Effect of Soil Regenerative Practice on Selected Soil Physical Properties and Eggplant (Solanum melongena L.) Yield. Agronomy 12: 1686. https://doi.org/10.3390/agronomy12071686.
Akinnifesi, F.K., O.C. Ajayi, G. Sileshi, P.W. Chirwa, and J. Chinau. 2010. Fertiliser Trees for Sustainable Food Security in the Maize-Based Production Systems of East and Southern Africa. A Review. Agronomy for Sustainable Development 30: 615–629. http://dx.doi.org/10.1051/agro/2009058.
Albaho, M.S., and J.L. Green. 2000. Suaeda salsa, a Desalinating Companion Plant for Greenhouse Tomato. HortScience 35(4): 620–623.
Ameline, A., J. Dorland, P.Y. Werrie, A. Couty, M.L. Fauconnier, M. Lateur, and G. Doury. 2022. Geranium macrorrhizum, a Potential Novel Companion Plant Affecting Preference and Performance of Myzus persicae on Sweet Pepper. Journal of Pest Science 96(2): 671–682.
Amoabeng, B.W., A.C. Johnson, and G.M. Gurr. 2019. Natural Enemy Enhancement and Botanical Insecticide Source: A Review of Dual Use Companion Plants. Applied Entomology and Zoology. https://doi.org/10.1007/s13355-018-00602-0.
Balmer, O., L. Pfiffner, J. Schied, M. Willareth, A. Leimgruber, H. Luka, and M. Traugott. 2013. Noncrop Flowering Plants Restore Top-Down Herbivore Control in Agricultural Fields. Ecology and Evolution 3(8): 2634–2646.
Balzan, M.V. 2017. Flowering Banker Plants for the Delivery of Multiple Agroecosystem Services. Arthropod-Plant Interactions 11(6): 743–754.
Borges, M., M.F.F. Michereff, R.A. Laumann, G.T. Santana, B.S. Castro, C.C. Silva, and M.C. Blassioli-Moraes. 2022. Influence of Pigeon Pea (Cajanus cajan) on Oviposition Behaviour of Diceraeus melacanthus Stink Bug, an Important Pest of Soybean and Maize Crops in South America. Arthropod – Plant Interactions 17(1): 77–89.
Breitenmoser, S., T. Steinger, A. Baux, and I. Hiltpold. 2022. Intercropping Winter Oilseed Rape (Brassica napus L.) Has the Potential to Lessen the Impact of the Insect Pest Complex. Agronomy 12: 723. https://doi.org/10.3390/agronomy12030723.
Chailleux, A., S. Ndjiliw, M. Diakhaté, G. Falone Akodjetin, P. Correa, E. Deletre, and T. Brévault. 2022. Approaches to Conservation of Nesidiocoris tenuis for Biological Control of Pests in Field-Grown Tomato in Senegal. Biological Control 172.
Chalker-Scott, L. 2013. The Science behind Biodynamic Preparations: A Literature Review. HortTechnology 23(6): 814–819.
Chalker-Scott, L. 2015a. Nonnative, Noninvasive Woody Species Can Enhance Urban Landscape Biodiversity. Arboriculture and Urban Forestry 41(4): 173–186.
Chalker-Scott, L. 2015b. Using Arborist Wood Chips as a Landscape Mulch. Washington State University Extension Publication FS160E. Washington State University.
Chalker-Scott, L. 2017. A Gardener’s Primer to Mycorrhizae: Understanding How They Work and Learning How to Protect Them. Washington State University Extension Publication FS269E. Washington State University.
Chalker-Scott, L. 2018. Are Native Trees and Shrubs Better Choices for Wildlife in Home Landscapes? Washington State University Extension Publication FS300E. Washington State University.
Close, D.C., N.J. Davidson, K.C. Churchill, and P. Grosser. 2005. Evaluation of Establishment Techniques on Eucalyptus nitens and E. pauciflora in the Midlands of Tasmania. Ecological Management and Restoration 6(2): 149–151.
Colla, G., Y. Rouphael, C. Fallovo, M. Cardarelli, and A. Graifenberg. 2006. Use of Salsola soda as a Companion Plant to Improve Greenhouse Pepper (Capsicum annuum) Performance under Saline Conditions. New Zealand Journal of Crop and Horticultural Science 34(4): 283–290.
Collier, R.H., and S. Finch. 2003. The Effect of Increased Crop Diversity on Colonisation by Pest Insects of Brassica Crops, Volumes 1 and 2. Proceedings of the BCPC International Congress: Crop Science and Technology, Glasgow, Scotland, UK, pp. 439–444.
Conboy, N.J.A., T. McDaniel, A. Ormerod, D. George, A.M.R. Gatehouse, E. Wharton, P. Donohoe, R. Curtis, and C.R. Tosh. 2019. Companion Planting with French Marigolds Protects Tomato Plants from Glasshouse Whiteflies through the Emission of Airborne Limonene. PLoS ONE. https://doi.org/10.1371/journal.pone.0213071.
Dardouri, T., L. Gomez, A. Ameline, G. Costagliola, A. Schoeny, and H. Gautier. 2021. Non-host Volatiles Disturb the Feeding Behavior and Reduce the Fecundity of the Green Peach Aphid, Myzus persicae. Pest Management Science 77(4): 1705–1713.
Dieudonné, E., H. Gautier, T. Dardouri, M. Staudt, G. Costagliola, and L. Gomez. 2022. Establishing Repellent Effects of Aromatic Companion Plants on Dysaphis plantaginea, Using a New Dynamic Tubular Olfactometer. Entomologia Experimentalis et Applicata 170(8): 727–743.
Finch, S., H. Billiald, and R.H. Collier. 2003. Companion Planting—Do Aromatic Plants Disrupt Host-Plant Finding by the Cabbage Root Fly and the Onion Fly More Effectively Than Non-aromatic Plants? Entomologia Experimentalis et Applicata 109(3): 183–195.
Finch, S., and R.H. Collier. 2000. Host-Plant Selection by Insects—A Theory Based on ‘Appropriate/Inappropriate Landings’ by Pest Insects of Cruciferous Plants. Entomologia Experimentalis et Applicata 96(2): 91–102.
Finch, S., and R.H. Collier. 2012. The Influence of Host and Non-host Companion Plants on the Behaviour of Pest Insects in Field Crops. Entomologia Experimentalis et Applicata 142(2): 87–96.
Fustec, J., F. Lesuffleur, S. Mahieu, and J.B. Cliquet. 2010. Nitrogen Rhizodeposition of Legumes. A Review. Agronomy for Sustainable Development 30(1): 57–66.
George, D., G. Port, and R. Collier. 2019. Living on the Edge: Using and Improving Trap Crops for Flea Beetle Management in Small-Scale Cropping Systems. Insects 10(9). DOI: https://doi.org/10.3390/insects10090286.
Gontijo, L.M., A.V. Saldanha, D.R. Souza, R.S. Viana, B.C. Bordin, and A.C. Antonio. 2018. Intercropping Hampers the Nocturnal Biological Control of Aphids. Annals of Applied Biology 172: 148–159.
Gospodarek, J. 2021. Effect of Sinapis alba L. as an Insectary Plant on the Occurrence of Aphis fabae Scop., Coccinellidae and Syrphidae in Broad Bean. Agronomy 11(11).
Graifenberg, A., L. Botrini, L. Giustiniani, F. Filippi, and M. Curadi. 2003. Tomato Growing in Saline Conditions with Biodesalinating Plants: Salsola soda L., and Portulaca oleracea L. Acta Horticulturae 609: 301–305.
Green, J.L., B. Blackburn, S. Kelly, and M. Albaho. 2000. Efficient Fertilization of Nursery Crops—Plant Controlled Uptake. Acta Horticulturae 511: 59–64.
Griffiths-Lee, J., E. Nicholls, and D. Goulson. 2020. Companion Planting to Attract Pollinators Increases the Yield and Quality of Strawberry Fruit in Gardens and Allotments. Ecological Entomology 45(5).
Hata, F.T., M.U. Ventura, M.T de Paula, G.D. Shimizu, J.C.B. de Paula, D.A.O. Kussaba, and N.V. de Souza. 2019. Intercropping Garlic in Strawberry Fields Improves Land Equivalent Ratio and Gross Income. Ciencia Rural 49(12). DOI: https://dx.doi.org/10.1590/0103-8478cr20190338.
Hatt, S., Q.X. Xu, F. Francis, and N. Osawa. 2019. Aromatic Plants of East Asia to Enhance Natural Enemies towards Biological Control of Insect Pests. A Review. Entomologia Generalis 38(4): 275–315. DOI: http://dx.doi.org/10.1127/entomologia/2019/0625.
Hemenway, T. 2001. Gaia’s Garden—A Guide to Home-Scale Permaculture. Chelsea Green Publishing Company, White River Junction, VT.
Holmgren, D. 2002. Permaculture: Principles and Pathways Beyond Sustainability. Holmgren Design Services, Hepburn, Victoria, Australia.
Jeavons, J. 2012. How to Grow More Vegetables, 8th edition. Ten Speed Press.
Kapayou, D.G., E.M. Herrighty, C.G. Hill, V.C. Camacho, A. Nair, D.M. Winham, and M.D. McDaniel. 2023. Reuniting the Three Sisters: Collaborative Science with Native Growers to Improve Soil and Community Health. Agriculture and Human Values 40(1): 65–82. DOI: https://doi.org/10.1007/s10460-022-10336-z.
Karakas, S., I. Bolat, and M. Dikilitas. 2021. The Use of Halophytic Companion Plant (Portulaca oleracea L.) on Some Growth, Fruit, and Biochemical Parameters of Strawberry Plants under Salt Stress. Horticulturae 7(4): 63.
Karakas, S., M.A. Cullu, C. Kaya, and M. Dikilitas. 2016. Halophytic Companion Plants Improve Growth and Physiological Parameters of Tomato Plants Grown under Salinity. Pakistan Journal of Botany 48(1): 21–28.
Khanal, U., K.J. Stott, R. Armstrong, J.G. Nuttall, F. Henry, B.P. Christy, M. Mitchell, et al. 2021. Intercropping—Evaluating the Advantages to Broadacre Systems. Agriculture 11: 453. DOI: https://doi.org/10.3390/agriculture11050453.
Kellogg, J.A., J.P. Reganold, K.M. Murphy, and L.A. Carpenter-Boggs. 2021. A Plant-Fungus Bioassay Supports the Classification of Quinoa (Chenopodium quinoa Willd.) as Inconsistently Mycorrhizal. Microbial Ecology 82(1): 135–144. DOI: http://dx.doi.org/10.1007/s00248-021-01710-1.
Laffon, L., A. Bischoff, H. Gautier, F. Gilles, L. Gomez, F. Lescourret, and P. Franck. 2022. Conservation Biological Control of Codling Moth (Cydia pomonella): Effects of Two Aromatic Plants, Basil (Ocimum basilicum) and French Marigolds (Tagetes patula). Insects 13: 908. https://doi.org/10.3390/insects13100908.
LaMondia, J.A., W.H. Elmer, T.L. Mervosh, and R.S. Cowles. 2002. Integrated Management of Strawberry Pests by Rotation and Intercropping. Crop Protection 21(9): 837–846.
Lérault, L., E. Clavel, C.M. Villegas, N. Cabrera, B. Jaloux, M. Plantegenest, and B. Lavandero. 2021. Providing Alternative Hosts and Nectar to Aphid Parasitoids in a Plum Orchard to Determine Resource Complementarity and Distance Range Effect on Biological Control. Agronomy 12: 77. https://doi.org/10.3390/agronomy12010077.
Li, S., C.C. Jaworski, S. Hatt, F. Zhang, N. Desneux, and S. Wang. 2020. Flower Strips Adjacent to Greenhouses Help Reduce Pest Populations and Insecticide Applications inside Organic Commercial Greenhouses. Journal of Pest Science 94: 679–689. DOI: http://dx.doi.org/10.1007/s10340-020-01285-9.
Liang, Y., C. Xu, H. Dai, J. Wang, X. Guo, S. Wang, and C.C. Jaworski. 2022. Flower Provision Reduces Intraguild Predation between Predators and Increases Aphid Biocontrol in Tomato. Journal of Pest Science 95: 461–472. https://doi.org/10.1007/s10340-021-01396-x.
Lopez, L., and O.E. Liburd. 2022. Can the Introduction of Companion Plants Increase Biological Control Services of Key Pests in Organic Squash? Entomologia Experimentalis et Applicata 170: 402–418. https://doi.org/10.1111/eea.13147.
Mansion-Vaquie, A., A. Ferrer, F. Ramon-Portugal, A. Wezel, and A. Magro. 2020. Intercropping Impacts the Host Location Behaviour and Population Growth of Aphids. Entomologia Experimentalis et Applicata 168(1): 41–52. DOI: http://dx.doi.org/10.1111/eea.12848.
Martins, D., R. Collier, and S. Finch. 2009. Host-Plant Finding by the Cabbage Root Fly Delia radicum L. Bulletin OIBC/OILB 51: 9–16.
McGee, P.A. 1990. Survival and Growth of Seedlings of Coachwood (Ceratopetalum apetalum): Effects of Shade, Mycorrhizas and a Companion Plant. Australian Journal of Botany 38(6): 583–592.
Milne, C., S.J. Trueman, S.H. Bai, and A. Shapcott. 2021. Translocation and Population Establishment of Schoenus scabripes (Cyperaceae). Australian Journal of Botany 69(4): 225–236.
Mollison, B. 1994. Introduction to Permaculture, 2nd edition. Tagari Publications, Australia.
Monti, M., A. Pellicanò, C. Santonoceto, G. Preiti, and A. Pristeri. 2016. Yield Components and Nitrogen Use in Cereal-Pea Intercrops in Mediterranean Environment. Field Crops Research 196: 379–388.
Montoya, J.E., Jr., M.A. Arnold, J. Rangel, L.R. Stein, and M.A. Palma. 2020. Pollinator-Attracting Companion Plantings Increase Crop Yield of Cucumbers and Habanero Peppers. HortScience 55(2): 164–169.
Mt. Pleasant, J., and R.F. Burt. 2010. Estimating Productivity of Traditional Iroquoian Cropping Systems from Field Experiments and Historical Literature. Journal of Ethnobiology 30(1): 52–79. DOI: https://doi.org/10.2993/0278-0771-30.1.52.
Ngapo, T.M., P. Bilodeau, Y. Arcand, M.T. Charles, A. Diederichsen, I. Germain, Q. Liu, et al. 2021. Historical Indigenous Food Preparation Using Produce of the Three Sisters Intercropping System. Foods 10(3): 524. DOI: https://doi.org/10.3390/foods10030524.
Nottingham, S.F. 1988. Host-Plant Finding by the Cabbage Root Fly, Delia radicum. Journal of Insect Physiology 34(3): 227–234.
Nyalala, S., and B. Grout. 2007. African Spider Flower (Cleome gynandra L./Gynandropsis gynandra (L.) Briq.) as a Red Spider Mite (Tetranychus urticae Koch) Repellent in Cut-Flower Rose (Rosa hybrida L.) Cultivation. Scientia Horticulturae 114(3): 194–198.
Otu, O.I., and A.A. Agboola. 1994. The Suitability of Gliricidia sepium In-Situ Live Stake on the Yield and Performance of White Yam (Dioscorea rotundata). Acta Horticulturae 380: 360–366.
Pålsson, J., M. Porcel, M.F. Hansen, J. Offenberg, T. Nardin, R. Larcher, and M. Tasin. 2020. Aphid-Infested Beans Divert Ant Attendance from the Rosy Apple Aphid in Apple-Bean Intercropping. Scientific Reports 10(5). DOI: https://doi.org/10.1038/s41598-020-64973-7.
Paula, R.R., J.-P. Bouillet, P.C.O Trivelin, B. Zeller, J.L. de M. Gonçalves, Y. Nouvellon, J.-M. Bouvet, C. Plassard, and J.-P. Laclau. 2015. Evidence of Short-term Belowground Transfer of Nitrogen from Acacia mangium to Eucalyptus grandis Trees in a Tropical Forest. Soil Biology & Biochemistry 91: 99–108.
Philbrick, H., and R.B. Gregg. 1966. Companion Plants and How to Use Them. The Devin-Adair Company, New York, NY.
Pouët, C., E. Deletre, and B. Rhino. 2021. Repellency of Wild Oregano Plant Volatiles, Plectranthus amboinicus, and Their Essential Oils to the Silverleaf Whitefly, Bemisia tabaci, on Tomato. Neotropical Entomology 51(1): 133–142.
Rascio, N., and F. Navari-Izzo. 2011. Heavy Metal Hyperaccumulating Plants: How and Why Do They Do It? And What Makes Them So Interesting? Plant Science 180: 169–181.
Riotte, L. 2004. Carrots Love Tomatoes: Secrets of Companion Planting for Successful Gardening. Storey Publishing, Pownal, VT.
Saldanha, A.V., L.M. Gontijo, R.M.R. Carvalho, C.J. Vasconcelos, A.S. Corrêa, and R.L.R. Gandra. 2019. Companion Planting Enhances Pest Suppression Despite Reducing Parasitoid Emergence. Basic and Applied Ecology 41: 45–55 DOI: https://doi.org/10.1016/j.baae.2019.10.002.
Sutter, L., V. Dekumbis, D. Maret, A. Ançay, and B. Christ. 2022. Floral Enhancements and UV-Mulch Tend to Reduce the Number of Flower Thrips and Their Associated Damage in Intensive Strawberry Production. IOBC/WPRS Bulletin 156: 6–10.
Tedersoo, L., L. Laanisto, S. Rahimlou, A. Toussaint, T. Hillikma, and M. Partel. 2018. Global Database of Plants with Root-Symbiotic Nitrogen Fixation: NodDB. Journal of Vegetation Science 29: 560–568.
Urli, M., N. Thiffault, D. Houle, S. Gauthier, and Y. Bergeron. 2020. Role of Green Alder in Boreal Conifer Growth: Competitor or Facilitator? Facets 5: 166–181. DOI: http://dx.doi.org/10.1139/facets-2019-0064.
Waiganjo, M.M., J. Muriuki, and G.W. Mbugua. 2007. Potential of Indigenous Leafy Vegetables as Companion Crops for Pest Management of High-Value Legumes: A Case Study of Gynandropsis gynandra in Kenya. Acta Horticulturae 752: 319–321.
Wang, Y., F. Yao, M.A. Soares, S.E. Basiri, E. Amiens-Desneux, M.R. Campos, A.-V. Lavoir, and N. Desneux. 2020. Effects of Four Non-Crop Plants on Life History Traits of the Lady Beetle Harmonia axyrid. Entomologia Generalis 40(3): 243–252.
Waweru, B.W., P. Rukundo, D.C. Kilalo, D.W. Miano, and J.W. Kimenju. 2021. Effect of Border Crops and Intercropping on Aphid Infestation and the Associated Viral Diseases in Hot Pepper (Capsicum sp.). Crop Protection 145: 105623. DOI: https://doi.org/10.1016/j.cropro.2021.105623.
Wikipedia. 2022. Land Equivalent Ratio. https://en.wikipedia.org/wiki/Land_equivalent_ratio.
Zhang, C.C., J.A. Postma, L.M. York, and J.P. Lynch. 2014. Root Foraging Elicits Niche Complementarity-Dependent Yield Advantage in the Ancient ‘Three Sisters’ (Maize/Bean/Squash) Polyculture. Annals of Botany 114(8): 1719–1733.
Zuma, M., C. Njekete, K.A.J. Konan, P. Bearez, E. Amiens-Desneux, N. Desneux, and A.V. Lavoir. 2022. Companion Plants and Alternative Prey Improve Biological Control by Orius laevigatus on Strawberry. Journal of Pest Science 96(2): 711–721.