Propagating Shrubs, Vines, and Trees from Stem Cuttings
Factors Affecting Adventitious Root Formation
All types of stem cuttings contain buds in the leaf axils (axillary or lateral buds), each of which can develop shoot systems. Survival and development of stem cuttings depend largely on the growth of adventitious roots to absorb water and nutrients. This form of development contrasts with the growth of primary roots from the radicle of germinating seeds. The main objective of propagating plants via stem cuttings is to facilitate adventitious root development.
Several plant-associated and environmental factors influence regeneration of roots from stem cuttings. Understanding the effects of these factors individually and in combination will help you achieve successful plant multiplication from cuttings.
Plant-Associated Factors
Plant species
Considerable variation in rooting ability exists among different plant species as well as the same species. The Propagation Reference Guide specifies some of these variations. For example, willows are considered easy to root because they have root initials (specialized cell groups) that readily develop into roots (Figure 1A). Hard-to-root plants may not root even with the application of root-inducing synthetic auxin (hormones).
Age of stock plant
In general, the ability of plants to develop adventitious roots declines with advancing age. Stem cuttings taken from young plants still in a juvenile stage of development root better than plants that have entered the reproductive (flowering) phase because of a difference in hormones or formation of root-inhibiting substances. The process of flowering also competes with rooting for the limited carbohydrate reserves in the stem cuttings (Figure 1C). However, cuttings taken from mature plants will likely root if they are an easy-to-root species (see the Propagation Reference Guide), their flower buds are removed (Figure 1B), their stems are treated with rooting hormone, or a combination of these factors.

Polarity
Stem cuttings always root at their proximal ends (i.e., toward the ground in relation to where on the mother or stock plant the cutting was removed) because of a dependence on natural auxin transport. Cuttings will not root if planted with their polarity (natural direction of growth) reversed.
Polarity can be easily determined for plants with buds above the leaf base by identifying the leaf bud direction on the stem cutting. Regardless, it is a good practice to identify polarity at the time the cuttings are taken. A slanted cut at the proximal end of cuttings (Figure 2) can be very useful in identifying polarity. A slanted cut also exposes a larger wounded area for hormone treatment and root development.

Presence or absence of leaves
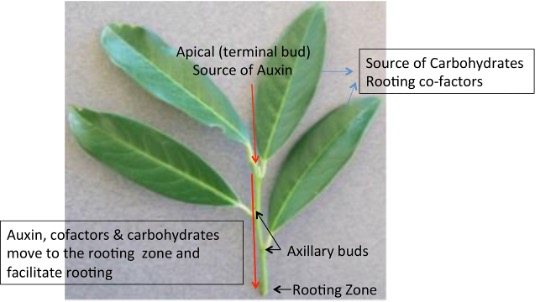
Retaining the leaves on cuttings helps roots develop because it provides carbohydrates, auxin, and rooting co-factors (Figure 3). However, cuttings that have large leaf areas require high relative humidity (such as misting) to avoid drying out. Reducing leaf area (either by rolling or partial removal of leaves; Figure 4) and using an improvised humidity chamber or propagation box also minimize moisture loss from stem cuttings (Figure 5A–E).


Nutrition of stock plant
Applying too much or too little fertilizer to stock plants may hinder the rooting ability of stem cuttings because it affects the plant’s growth and nutritional balance. Follow the general recommendations for the plant you are growing. Nitrogen in particular plays a decisive role in rooting stock plants. Exceeding recommended levels of nitrogen may cause succulent stems in the stock plant. Stem cuttings taken from such plants tend to rot. If your source plant shows signs of succulent growth, reduce nitrogen fertilization.
Wounding
Stem wounding (a cut in the stem) encourages adventitious root formation (Figure 6A–C) because it activates cell division in the tissue between the bark and wood. Wounding also helps plants absorb water and rooting hormones (Figure 6C) and breaks through structural tissue that interferes with root emergence.
The wounding that occurs during the preparation of stem cuttings is sufficient to induce adventitious root formation in many plant species, though additional wounding followed by the application of rooting hormone is often needed for hard-to-root plant species. Recommended methods include 1) removing a thin slice of bark at the base of the stem cutting (on one or both sides; Figure 6A) with a knife, 2) stripping off the lower leaves or branches of the stem cutting, or 3) splitting the basal end of the cutting by 0.5–1 inches (Figure 6C).

Synthetic growth regulators
Indole-3-butyric acid (IBA) and naphthaleneacetic acid (NAA) are growth regulators commonly used to regenerate roots on stem cuttings. Both are available in liquid and powder (talc) formulations that contain different concentrations of rooting hormones. The powder formulations are the most practical for propagators. Talc, which is used as a carrier, is inert and has no effect on rooting by itself. Common commercial root-promoting formulations available at local nursery, garden, or variety stores are Rootone, Hormex, and Hormodin.
You can also make your own rooting formulation with talc, reagent-grade rooting hormone (IBA or NAA), and isopropyl alcohol. First, dissolve the required amount of rooting hormone (provided in the Propagation Reference Guide) in 5–10 teaspoons of alcohol and add enough talc to form a slurry. Then evaporate the alcohol using gentle heat. Incorporate any clumps to ensure an even consistency. Discard any leftover powder to prevent contamination. (Talc preparations lose their effectiveness after about eight months, regardless of refrigeration.)
Environmental Factors
Time of year
The shaded areas in the Propagation Reference Guide indicate the ideal time for taking stem cuttings, which is dictated by plant species and type of stem cutting (softwood, semi-hardwood, hardwood). Softwood cuttings are taken during the growing season from new growth, semi-hardwood cuttings are taken when a stem’s wood is partially matured (characterized by relatively rigid stems), and hardwood cuttings are taken from stems after the completion of the growing period.
Cuttings taken from many deciduous plants and narrow-leaved evergreens from late fall to early winter are likely to root well. At this stage, shoot growth has slowed down and the stem has begun to harden, resulting in the accumulation of adequate carbohydrate reserves to support root growth. The relatively lower nitrogen levels also make cuttings less susceptible to rotting. Such stem cuttings root under warm conditions (such as in a propagation box or greenhouse). After rooting, the cuttings must be subjected to the cold temperatures associated with plant-specific dormancy requirements. Dormancy (rest period) requirements for selected fruit crops are listed in publications by Westwood (1993), Childers et al. (1995), and Ryugo (1988).
Stem cuttings from deciduous plants can also successfully root when taken during the growing season at the softwood or semi-hardwood stage. Immediately after a flush of growth is the optimal time to take stem cuttings from broadleaf evergreen plants.
Temperature
Adventitious root initiation and development are influenced by temperatures at the root zone. The optimum temperature for rooting most temperate plants is between 65 and 77°F. Tropical and subtropical plants are generally acclimated to warmer temperatures and root better at somewhat higher temperatures (77–89°F). You can manipulate temperature at the root zone by placing flats containing stem cuttings on heating coils (or hot water tubes; Figure 7A). Heating pads manufactured for propagation (Figure 7B) are also effective.
Hardwood stem cuttings from some difficult-to-root deciduous plants root best in a moist medium with bottom heat at 60–70°F. As soon as you can see roots, lower the temperature of the rooting medium to approximately 40°F to complete the cold (rest) period the buds require. This will also delay further root growth until the cutting is ready for planting the following spring. Stem cuttings handled in this way are often tied in bundles with all the basal ends pointing in the same direction. Following storage, untie the bundles and plant individual cuttings outdoors in a closely spaced nursery row. Most leafy stem cuttings do best when the air temperature in the propagation box is 60–65°F.

Rooting media
An ideal rooting medium provides physical support and aeration and also holds sufficient moisture for the cuttings. Sand, vermiculite, perlite (Figure 8A–C), peat, or some combination of these materials is most commonly used, but very fine gravel will also work.

Media and container sterilization
Media and containers should be free of disease-causing microorganisms. For most media, this is best accomplished in an oven at 140°F for 30 minutes; heating is not needed when new perlite is used. Containers can be washed with a solution of 1 part bleach and 9 parts water to kill microorganisms.
Water
Cuttings should remain turgid (hydrated) for successful generation of roots. Because new stem cuttings do not have roots, they cannot absorb enough water to stay alive. In addition, transpiration (water loss from leaves) contributes to desiccation. Commercial growers use intermittent misting to maintain a continuous film of moisture on plant leaves (Figure 7A). This mist lowers leaf temperature, creates a humid environment, and reduces water loss through transpiration. For most home gardeners, misting equipment is not economical, but relative humidity can be maintained in an improvised humidity chamber (Figure 5) or a wooden propagation box with a glass or clear plastic lid that admits light. Refer to Christiano (2013) for guidance if you intend to build a propagation box with bottom heat.
A propagation box keeps the air surrounding cuttings humid, but it cannot compensate for the cooling effect of misting. Place the box in an area that does not get direct sunlight to avoid heat buildup. Daily use of a spray bottle or mister may be needed for adequate humidity. Keep the rooting medium moist. Provide drainage in the bottom of the container to avoid waterlogging, which leads to decay of plant tissue. Often, deciduous hardwood stem cuttings can be rooted without mist or even the protection of a covered propagation box. For sustaining cuttings that take a long time to root or develop leaves prior to rooting, a propagation box is beneficial.
Light
Although light promotes production of carbohydrates in leaf-bearing cuttings, direct exposure to sunlight should be avoided to minimize water loss.
Preparing Cuttings
- Softwood: Use 3–5 inches of soft tissue with at least two nodes. Remove the lower leaves.
- Semi-hardwood: Use the top 4–6 inches of partially mature stems and keep the leaves intact.
- Hardwood: Use 2–30 inches of well-matured, hard parts of stems. Remove all leaves.
- Tip: Use the top 4–10 inches of main shoots (Figure 9A) or side branches of stems that are about one year old; cuttings from either a main shoot or side stem should include the uppermost bud.
- Heel: Use either the main stem (Figure 9B) or side branches of stems that are at least two years old; the length can vary. Remove the branches from the main stem, along with a piece of bark (called the heel).
- Mallet: Use a complete cross-section of the main stem, along with a side shoot (Figure 9C).

Planting Cuttings
- Treat cuttings with a rooting powder formulation.
- Make shallow holes in the rooting medium with a dowel, pencil, or stick.
- If you plan to use bottom heat, insert the base of the cuttings near the bottom of the propagation box so they are close enough to the heat source.
- Firm the rooting medium around the base of each cutting.
- Apply sufficient water to settle the rooting medium around the cuttings and cover the container with a poly film or place it under mist.
- Inspect the cuttings daily and remove dropped leaves.
- Keep the rooting medium moist. When the cuttings resist a slight tug and begin to feel anchored, they are beginning to root. The time required for rooting varies considerably according to the plant species. For example, rhododendrons may require three months or more to form sufficient roots to allow removal from a rooting medium.
- Once the cuttings have 2–3 roots about 0.5 inches long, place them in pots about 4 inches in diameter filled with any commercially available potting mix.
- Acclimate the individually potted cuttings to the temperature and humidity outside the propagation box by covering them with perforated plastic film for approximately one week. Keep them away from direct sunlight and temperature extremes for several weeks. This acclimatizing process is known as hardening. It can also be started before the cuttings are potted (between planting steps 7 and 8) by gradually aerating the propagation box to reduce the humidity.
- Transplant the cuttings outdoors in the spring when temperatures remain above freezing.
Exceptions to the General Planting Process
Some hardwood deciduous stem cuttings do not require a propagation box or misting. Those taken from easy-to-root plant types in the fall can be directly rooted in the soil outdoors where winter temperatures do not drop below 50°F. Cuttings from difficult-to-root plant types perform better when taken in the late fall.
Glossary
adventitious roots. Roots developing from plant parts, such as stems or leaves, as opposed to the primary root that develops from the radicle of germinating seeds.
asexual propagation. Regeneration of plants from vegetative parts of a plant, such as stems, leaves, or roots. Also called vegetative propagation.
auxin. A group of plant hormones with varied functions, such as cell elongation and adventitious root formation. Synthetic auxins, or growth regulators such as IBA and NAA, are commonly used to initiate adventitious roots on cuttings.
clones. Genetically identical plants developed from the vegetative parts of a single source plant.
deciduous. Characteristic of plants that become dormant and lose leaves in the fall in response to a drop in temperature.
evergreen. Characteristic of plants that retain leaves year round.
hardening. A process that prepares cuttings to withstand temperature and humidity conditions outside a propagation box or greenhouse. Also called acclimating.
hardwood cutting. A woody piece of stem obtained any time from fall through late winter.
juvenile stage. Developmental period of a plant characterized by vegetative growth. Cuttings taken from plants during this phase generally root well.
leaf axil. The point where a leaf is attached to the main stem at an angle. Generally, vegetative or floral buds originate at leaf axils. Also called axillary or lateral buds.
nodes. Hardened parts on a plant stem where buds and leaves grow.
polarity. The natural direction a plant grows, which is dictated by the internal production and transport of auxin.
radicle. Part of the embryo in a seed that develops into a root during germination.
reagent-grade. The purest form of chemicals available commercially.
rooting co-factors. Compounds produced by plants that work synergistically with auxins to facilitate adventitious root formation.
root initials. A group of cells capable of forming roots.
semi-hardwood cutting. Stem cuttings that are relatively less turgid and stiff than hardwood cuttings.
softwood cutting. Stem cuttings that are turgid and tender.
stem cutting. A piece of plant stem with axillary buds, terminal buds, or both used in plant multiplication.
succulent stems. Stems characterized by soft tissue and excessive water accumulation. Excessive nitrogen fertilization can be a contributory factor. Cuttings taken from such stems seldom root well.
transpiration. Loss of water in the form of water vapor from leaf surfaces.
turgid. A condition when plant organs, such as leaves, have reached full hydration. Turgid plants have well-expanded leaves and erect stems. Loss of turgidity leads to wilting and then desiccation.
Acknowledgements
This publication is a revision of:
Larsen, F.E. 1997. Propagating Deciduous and Evergreen Shrubs, Trees, and Vines with Stem Cuttings. Pacific Northwest Extension Publication PNW152. Washington State University.
The author thanks Linda McMahan, Oregon State University, and Bob Tripepi, University of Idaho, for their constructive peer reviews, and Dora Rollins, Christina Mangiapani, and Lagene Taylor, Washington State University, for editing and coordinating the publication of this manuscript.
Further Reading
Bryant, G. 2006. Plant Propagation A to Z: Growing Plants for Free. New York: Firefly Books.
Hartmann, H.T., D.E. Kester, F.T. Davies, and R.L. Geneve. 2011. Plant Propagation: Principles and Practices. 8th ed. New York: Prentice Hall.
Rose, R., C.E.C. Chachulski, and D.L. Haase. 1998. Propagation of Pacific Northwest Native Plants. Corvallis: Oregon State University Press.
Toogood, A. 1999. Plant Propagation: The Fully Illustrated Plant-by-Plant Manual of Practical Techniques. New York: DK Publishing.
References
Childers, N.F., J.R., Morris, and G.S. Sibbett. 1995. Modern Fruit Science: Orchard and Small Fruit Management.
Gainesville, FL: Horticultural Publications.
Christiano, D. 2013. Build a Long-Lasting Bottom-Heat Propagating Box.
Ryugo, K. 1988. Fruit Culture: It’s Science and Art. New York: John Wiley and Sons.
Westwood, M.N. 1993. Temperate-Zone Pomology: Physiology and Culture. 3rd ed. Portland, OR: Timber Press.
Published and distributed in furtherance of the Acts of Congress of May 8 and June 30, 1914, by Washington State University Extension, Oregon State University Extension Service, University of Idaho Extension, and the U.S. Department of Agriculture cooperating. WSU Extension programs, activities, materials, and policies comply with federal and state laws and regulations on nondiscrimination regarding race, sex, religion, age, color, creed, and national or ethnic origin; physical, mental, or sensory disability; marital status or sexual orientation; and status as a Vietnam-era or disabled veteran. Washington State University Extension, Oregon State University Extension Service, and University of Idaho Extension are Equal Opportunity Employers. Evidence of noncompliance may be reported through your local Extension office. Trade names have been used to simplify information; no endorsement is intended.
Pacific Northwest Extension publications contain material written and produced for public distribution. You may reprint written material, provided you do not use it to endorse a commercial product. Please reference by title and credit Pacific Northwest Extension publications.
Order Information:
WSU Extension
Fax: 509-335-3006
Toll-free phone: 800-723-1763
Email: ext.pubs@wsu.edu
OSU Extension
Fax: 541-737-0817
Toll-free phone: 800-561-6719
Email: puborders@oregonstate.edu
UI Extension
Fax: 208-885-4648
Phone: 208-885-7982
Email: calspubs@uidaho.edu
Copyright © Washington State University
Pacific Northwest Extension publications are produced cooperatively by the three Pacific Northwest land-grant universities: Washington State University, Oregon State University, and the University of Idaho. Similar crops, climate, and topography create a natural geographic unit that crosses state lines. Since 1949, the PNW program has published more than 650 titles, preventing duplication of effort, broadening the availability of faculty specialists, and substantially reducing costs for the participating states.




